Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
Pages: 1
#1 2026-03-29 00:01:51
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,813
Spectroscopy
Spectroscopy
Gist
Spectroscopy is the study of the interaction between matter and electromagnetic radiation, analyzing how substances absorb or emit light to identify their composition, structure, and physical properties. By analyzing spectral "fingerprints," this technique is critical in chemistry, astronomy, medicine (e.g., MRI), and material science to determine molecular structures and elemental composition.
Spectroscopy is the study of how matter interacts with electromagnetic radiation (light, UV, infrared, X-rays). It measures the absorption, emission, or scattering of radiation by substances, creating a unique spectrum that serves as a "fingerprint" to identify chemical composition, structure, and physical properties.
Summary
Spectroscopy is the study of the interaction between matter and electromagnetic radiation. This can either involve the absorption of radiation by the sample, or the emission of radiation from the sample. In a typically emission spectrum, the variation of the intensity of the emitted radiation with frequency is measured. In the Department, we use and study a wide range of spectroscopic techniques, which give information about a huge number of different materials. We also study the theory of spectroscopy computationally.
Some spectroscopy techniques access the average or typical properties of a material, whilst others are employed inside a microscope and can give very local information. Examples of spectroscopy techniques used in a transmission electron microscope include electron energy loss spectroscopy used to study the local compositions of materials, whilst in the scanning electron microscope we use cathodoluminescence spectroscopy to understand the light emission properties of semiconductors and other structures and in the scanning tunneling microscope spectroscopic techniques give insights into electronic structure. Solid state NMR provides a sensitive probe of material structure, and is sensitive to local order and the dynamics of the system.
Theoretical spectroscopy, typically based on first principles density functional theory (DFT) computations, can provide a link between the material structure and the spectra measured. These calculations can aid the design of experiments, or assist in structure solution.
Details
Spectroscopy is the field of study that measures and interprets electromagnetic spectra as it interacts with matter. In narrower contexts, spectroscopy is the precise study of color as generalized from radiated visible light to all bands of the electromagnetic spectrum.
Spectroscopy, primarily in the electromagnetic spectrum, is a fundamental exploratory tool in the fields of astronomy, chemistry, materials science, and physics, allowing the composition, physical and electronic structure of matter to be investigated at the atomic, molecular and macro scale, and over astronomical distances.
Historically, spectroscopy originated as the study of the wavelength dependence of the absorption by gas phase matter of visible light dispersed by a prism. Current applications of spectroscopy include biomedical spectroscopy in the areas of tissue analysis and medical imaging. Matter waves and acoustic waves can be considered forms of radiative energy, and recently gravitational waves have been associated with a spectral signature in the context of the Laser Interferometer Gravitational-Wave Observatory (LIGO).
Introduction
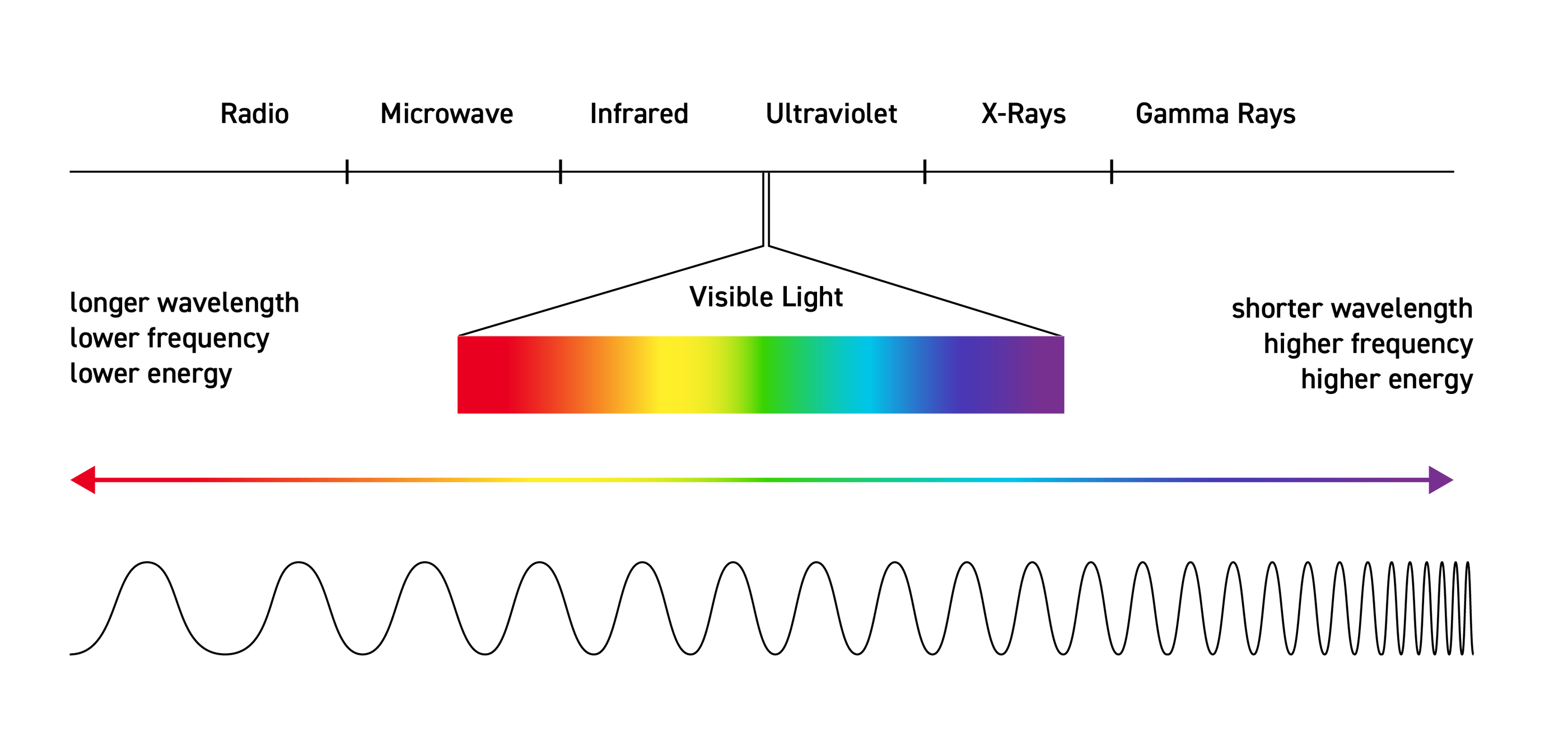
Spectroscopy is a branch of science concerned with the spectra of electromagnetic radiation as a function of its wavelength or frequency, as measured by spectrographic equipment and other techniques, in order to obtain information concerning the structure and properties of matter. Spectral measurement devices are referred to as spectrometers, spectrophotometers, spectrographs or spectral analyzers. Most spectroscopic analysis in the laboratory starts with a sample to be analyzed. A light source is sent through a monochromator to spatially separate the colors before passing a selected frequency band through the sample, then the output is captured by a photodiode. For astronomical purposes, the telescope must be equipped with the light dispersion device. There are various versions of this basic setup that may be employed.
Spectroscopy began with Isaac Newton splitting light with a prism; a key moment in the development of modern optics. Therefore, it was originally the study of visible light that we call color. Following the contributions of James Clerk Maxwell, this study later came to include the entire electromagnetic spectrum. Although color is involved in spectroscopy, it is not equivalent to the absorption and reflection of certain electromagnetic waves that give objects or elements a sense of color to our eyes. Rather, spectroscopy involves the splitting of light by a prism, diffraction grating, or similar instrument, to display a particular discrete line pattern called a "spectrum", which is unique for each different type of element or molecule. Most elements are first put into a gaseous state to allow the spectra to be examined, although today other methods can be used for different phases of matter. Each element that is diffracted by a prism-like instrument displays either an absorption spectrum or an emission spectrum depending upon whether the element is being cooled or heated.
Until recently all spectroscopy involved the study of line spectra and most spectroscopy still does. Vibrational spectroscopy is the branch of spectroscopy that studies the spectra, which are caused by vibrations of molecules. However, the latest developments in spectroscopy can sometimes dispense with the dispersion technique. In biochemical spectroscopy, information can be gathered about biological tissue by absorption and light scattering techniques. Light scattering spectroscopy is a type of reflectance spectroscopy that determines tissue structures by examining elastic scattering. In such a case, it is the tissue that acts as a diffraction or dispersion mechanism.
Spectroscopic studies were central to the development of quantum mechanics. The first useful quantum atomic models, including Bohr model, the Schrödinger equation, and Matrix mechanics, reproduced the spectral lines of hydrogen. These equated discrete quantum jumps of the bound electron in a hydrogen atom to the discrete hydrogen spectrum. Max Planck's explanation of blackbody radiation involved spectroscopy because he was comparing the wavelength of light using a photometer to the temperature of a Black Body. Spectroscopy is used in physical and analytical chemistry because atoms and molecules have unique spectra. As a result, these spectra can be used to detect, identify and quantify information about the atoms and molecules.
Spectroscopy is used in astronomy and remote sensing on Earth. Most research telescopes have spectrographs. The measured spectra are used to determine the chemical composition and physical properties of astronomical objects, such as their temperature, elemental abundances, velocity, rotation, magnetic field, and more. An important use for spectroscopy is in biochemistry. Molecular samples may be analyzed for species identification and energy content.
Additional Information
Spectroscopy is the study of the absorption and emission of light and other radiation by matter, as related to the dependence of these processes on the wavelength of the radiation. More recently, the definition has been expanded to include the study of the interactions between particles such as electrons, protons, and ions, as well as their interaction with other particles as a function of their collision energy. Spectroscopic analysis has been crucial in the development of the most fundamental theories in physics, including quantum mechanics, the special and general theories of relativity, and quantum electrodynamics. Spectroscopy, as applied to high-energy collisions, has been a key tool in developing scientific understanding not only of the electromagnetic force but also of the strong and weak nuclear forces.
Spectroscopic techniques have been applied in virtually all technical fields of science and technology. Radio-frequency spectroscopy of nuclei in a magnetic field has been employed in a medical technique called magnetic resonance imaging (MRI) to visualize the internal soft tissue of the body with unprecedented resolution. Microwave spectroscopy was used to discover the so-called three-degree blackbody radiation, the remnant of the big bang (i.e., the primeval explosion) from which the universe is thought to have originated (see below Survey of optical spectroscopy: General principles: Applications). The internal structure of the proton and neutron and the state of the early universe up to the first thousandth of a second of its existence are being unraveled with spectroscopic techniques using high-energy particle accelerators. The constituents of distant stars, intergalactic molecules, and even the primordial abundance of the elements before the formation of the first stars can be determined by optical, radio, and X-ray spectroscopy. Optical spectroscopy is used routinely to identify the chemical composition of matter and to determine its physical structure.
Spectroscopic techniques are extremely sensitive. Single atoms and even different isotopes of the same atom can be detected among {10}^{20} or more atoms of a different species. (Isotopes are all atoms of an element that have unequal mass but the same atomic number. Isotopes of the same element are virtually identical chemically.) Trace amounts of pollutants or contaminants are often detected most effectively by spectroscopic techniques. Certain types of microwave, optical, and gamma-ray spectroscopy are capable of measuring infinitesimal frequency shifts in narrow spectroscopic lines. Frequency shifts as small as one part in {10}^{15} of the frequency being measured can be observed with ultrahigh resolution laser techniques. Because of this sensitivity, the most accurate physical measurements have been frequency measurements.
Spectroscopy now covers a sizable fraction of the electromagnetic spectrum. The table summarizes the electromagnetic spectrum over a frequency range of 16 orders of magnitude. Spectroscopic techniques are not confined to electromagnetic radiation, however. Because the energy E of a photon (a quantum of light) is related to its frequency ν by the relation E = hν, where h is Planck’s constant, spectroscopy is actually the measure of the interaction of photons with matter as a function of the photon energy. In instances where the probe particle is not a photon, spectroscopy refers to the measurement of how the particle interacts with the test particle or material as a function of the energy of the probe particle.
An example of particle spectroscopy is a surface analysis technique known as electron energy loss spectroscopy (EELS) that measures the energy lost when low-energy electrons (typically 5–10 electron volts) collide with a surface. Occasionally, the colliding electron loses energy by exciting the surface; by measuring the electron’s energy loss, vibrational excitations associated with the surface can be measured. On the other end of the energy spectrum, if an electron collides with another particle at exceedingly high energies, a wealth of subatomic particles is produced. Most of what is known in particle physics (the study of subatomic particles) has been gained by analyzing the total particle production or the production of certain particles as a function of the incident energies of electrons and protons.
The following sections focus on the methods of electromagnetic spectroscopy, particularly optical spectroscopy. Although most of the other forms of spectroscopy are not covered in detail, they have the same common heritage as optical spectroscopy. Thus, many of the basic principles used in other spectroscopies share many of the general features of optical spectroscopy.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1