Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
#276 Re: Jai Ganesh's Puzzles » English language puzzles » 2026-02-26 15:36:01
Hi,
#5965. What does the noun harmony mean?
#5966. What does the noun harness mean?
#277 Re: Jai Ganesh's Puzzles » Doc, Doc! » 2026-02-26 15:24:51
Hi,
#2579. What does the medical term Cementum mean?
#278 Re: Jai Ganesh's Puzzles » Oral puzzles » 2026-02-26 14:52:32
Hi,
#6359.
#279 Re: Jai Ganesh's Puzzles » 10 second questions » 2026-02-26 14:27:12
Hi,
#9865.
#280 Re: Exercises » Compute the solution: » 2026-02-26 14:08:02
Hi,
2719.
#281 Re: This is Cool » Miscellany » 2026-02-26 00:55:40
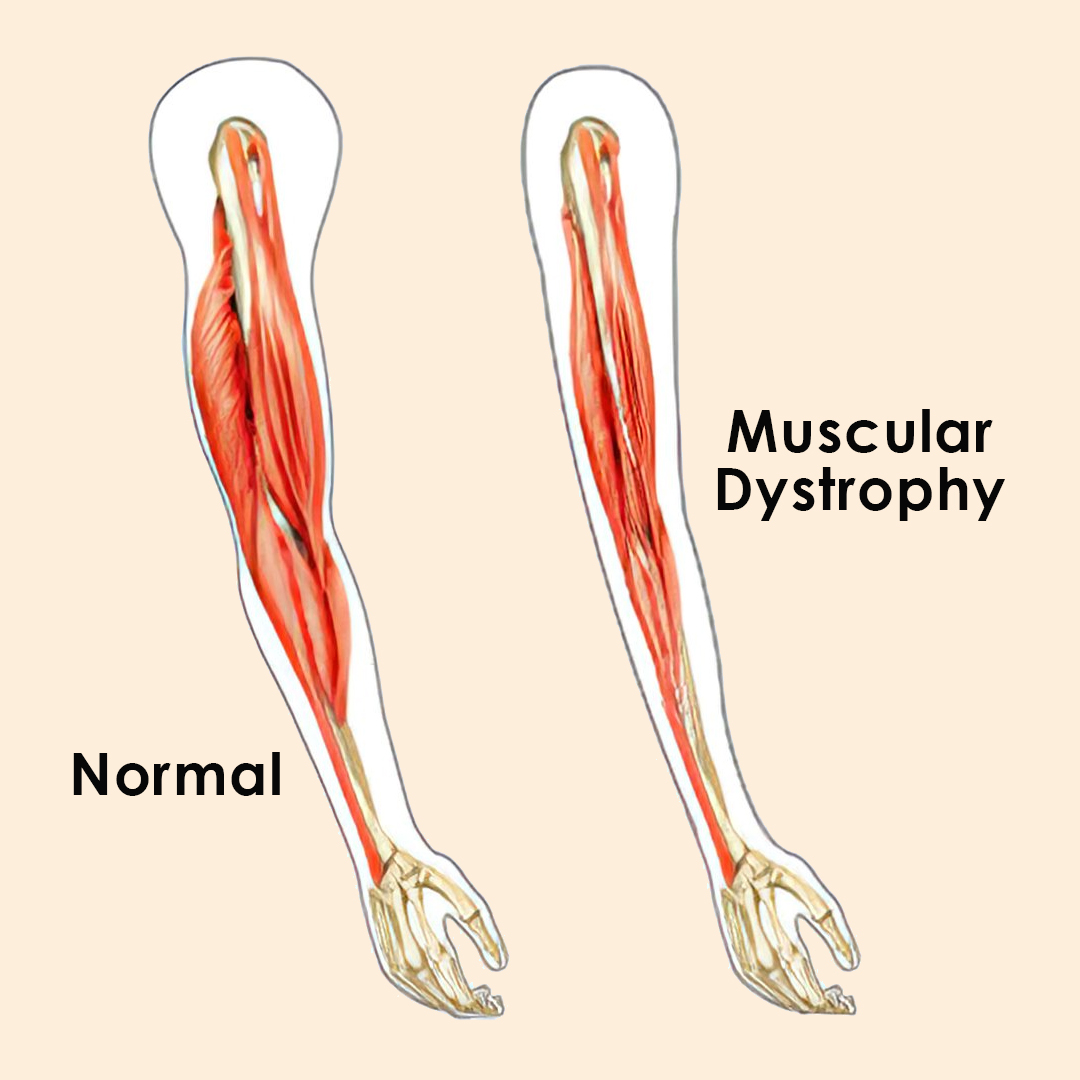
2505) Atrophy
Gist
Atrophy is the wasting away, thinning, or decrease in size of cells, tissues, or organs, resulting in reduced function. It is caused by factors like disuse, poor circulation, nerve damage, malnutrition, or hormonal changes. Common forms include muscle, brain, or vaginal atrophy, often characterized by weakness and reduced size of the affected area.
Brain atrophy means the progressive loss of brain cells (neurons) and their connections, causing the brain to shrink in overall size or in specific regions, leading to impaired cognitive, motor, or functional abilities, and can be a normal part of aging or a symptom of diseases like Alzheimer's or MS (Multiple Sclerosis). It's diagnosed via brain scans like MRIs and CTs, which reveal the tissue loss, and treatment focuses on managing the underlying cause.
Summary
Muscle atrophy is the wasting or thinning of muscle mass. It can be caused by disuse of your muscles or neurogenic conditions. Symptoms include a decrease in muscle mass, one limb being smaller than the other, and numbness, weakness and tingling in your limbs. Disuse atrophy can be reversed with exercise and a healthy diet.
Muscle atrophy is the loss or thinning of your muscle tissue. If you have atrophied muscles, you’ll see a decrease in your muscle mass and strength. With muscle atrophy, your muscles look smaller than normal. Muscle atrophy can occur due to malnutrition, age, genetics, a lack of physical activity or certain medical conditions. Disuse (physiologic) atrophy occurs when you don’t use your muscles enough. Neurogenic atrophy occurs due to nerve problems or diseases.
What are the symptoms of muscle atrophy?
The symptoms of muscle atrophy differ depending on the cause of your condition. The most obvious sign of muscle atrophy is reduced muscle mass. Other signs of muscle atrophy may include:
* One arm or one leg is smaller than the other.
* Weakness in one arm and or one leg.
* Numbness or tingling in your arms and legs.
* Trouble walking or balancing.
* Difficulty swallowing or speaking.
* Facial weakness.
* Gradual memory loss.
Details
Atrophy is the partial or complete wasting away of a part of the body. Causes of atrophy include mutations (which can destroy the gene to build up the organ), poor nourishment, poor circulation, loss of hormonal support, loss of nerve supply to the target organ, excessive amount of apoptosis of cells, and disuse or lack of exercise or disease intrinsic to the tissue itself. In medical practice, hormonal and nerve inputs that maintain an organ or body part are said to have trophic effects. A diminished muscular trophic condition is designated as atrophy. Atrophy is reduction in size of cell, organ or tissue, after attaining its normal mature growth. In contrast, hypoplasia is the reduction in the cellular numbers of an organ, or tissue that has not attained normal maturity.
Atrophy is the general physiological process of reabsorption and breakdown of tissues, involving apoptosis. When it occurs as a result of disease or loss of trophic support because of other diseases, it is termed pathological atrophy, although it can be a part of normal body development and homeostasis as well.
Examples of atrophy as part of normal development include shrinking and the involution of the thymus in early childhood, and the tonsils in adolescence. In old age, effects include, but are not limited to, loss of teeth, hair, thinning of skin that creates wrinkles, weakening of muscles, loss of weight in organs and sluggish mental activity.
Muscle atrophies
Disuse atrophy of muscles and bones, with loss of mass and strength, can occur after prolonged immobility, such as extended bedrest, or having a body part in a cast (living in darkness for the eye, bedridden for the legs etc.). This type of atrophy can usually be reversed with exercise unless severe.
There are many diseases and conditions which cause atrophy of muscle mass. For example, diseases such as cancer and AIDS induce a body wasting syndrome called cachexia, which is notable for the severe muscle atrophy seen. Other syndromes or conditions which can induce skeletal muscle atrophy are congestive heart failure and liver disease.
During aging, there is a gradual decrease in the ability to maintain skeletal muscle function and mass. This condition is called sarcopenia, and may be distinct from atrophy in its pathophysiology. While the exact cause of sarcopenia is unknown, it may be induced by a combination of a gradual failure in the satellite cells which help to regenerate skeletal muscle fibers, and a decrease in sensitivity to or the availability of critical secreted growth factors which are necessary to maintain muscle mass and satellite cell survival.
Dystrophies, myositis, and motor neuron conditions
Pathologic atrophy of muscles can occur with diseases of the motor nerves or diseases of the muscle tissue itself. Examples of atrophying nerve diseases include Charcot-Marie-Tooth disease, poliomyelitis, amyotrophic lateral sclerosis (ALS or Lou Gehrig's disease), and Guillain–Barré syndrome. Examples of atrophying muscle diseases include muscular dystrophy, myotonia congenita, and myotonic dystrophy.
Changes in Na+ channel isoform expression and spontaneous activity in muscle called fibrillation can also result in muscle atrophy.
A flail limb is a medical term which refers to an extremity in which the primary nerve has been severed, resulting in complete lack of mobility and sensation. The muscles soon wither away from atrophy.
Gland atrophy
The adrenal glands atrophy during prolonged use of exogenous glucocorticoids like prednisone. Atrophy of the breasts can occur with prolonged estrogen reduction, as with anorexia nervosa or menopause. Testicular atrophy can occur with prolonged use of enough exogenous sex steroids (either androgen or estrogen) to reduce gonadotropin secretion.
Vaginal atrophy
In post-menopausal women, the walls of the math become thinner (atrophic vaginitis). The mechanism for the age-related condition is not yet clear, though there are theories that the effect is caused by decreases in estrogen levels. This atrophy, occurring concurrently with breast atrophy, is consistent with the homeostatic (normal development) role of atrophy in general, as after menopause the body has no further functional biological need to maintain the reproductive system which it has permanently shut down.
Research
One drug in test seemed to prevent the type of muscle loss that occurs in immobile, bedridden patients. Testing on mice showed that it blocked the activity of a protein present in the muscle that is involved in muscle atrophy. However, the drug's long-term effect on the heart precludes its routine use in humans, and other drugs are being sought.
Additional Information
Atrophy is a decrease in size of a body part, cell, organ, or other tissue. The term implies that the atrophied part was of a size normal for the individual, considering age and circumstance, prior to the diminution. In atrophy of an organ or body part, there may be a reduction in the number or in the size of the component cells, or in both.
Certain cells and organs normally undergo atrophy at certain ages or under certain physiologic circumstances. In the human embryo, for example, a number of structures are transient and at birth have already undergone atrophy. The adrenal glands become smaller shortly after birth because an inner layer of the cortex has shrunk. The thymus and other lymphoid tissues atrophy at adolescence. The pineal gland tends to atrophy about the time of puberty; usually calcium deposits, or concretions, form in the atrophic tissue. The widespread atrophy of many tissues that accompanies advanced age, although universal, is influenced by changes of nutrition and blood supply that occur during active mature life.
The normal cyclic changes of female reproductive organs are accompanied by physiologic atrophy of portions of these organs. During the menstrual cycle, the corpus luteum of the ovary atrophies if pregnancy has not occurred. The muscles of the uterus, which enlarge during pregnancy, rapidly atrophy after the delivery of the child, and after completion of lactation the milk-producing acinar structures of the breast diminish in size. After menopause the ovaries, uterus, and breasts normally undergo a degree of atrophic change.
Whole body atrophy
Atrophy in general is related to changes in nutrition and metabolic activity of cells and tissues. A widespread or generalized atrophy of body tissues occurs under conditions of starvation, whether because food is unavailable or because it cannot be taken and absorbed because of the presence of disease. The unavailability of certain essential protein components and vitamins disturbs the metabolic processes and leads to atrophy of cells and tissues. Under conditions of protein starvation, the body protein is broken down into constituent amino acids, which serve to provide energy and help maintain the structure and cells of the most essential organs. The brain, heart, adrenal glands, thyroid gland, pituitary gland, gonads, and kidneys show less atrophy, relatively, than the body as a whole, whereas the fatty stores of the body, liver, spleen, and lymphoid tissues diminish relatively more than the body as a whole. The brain, heart, and kidneys, organs with abundant blood supply, appear to be the least subject to the wasting effects of starvation.
Associated with the widespread atrophy due to lack of protein is the atrophy of certain tissues that is caused by deficiencies of specific vitamins. Atrophic changes of the skin increase because of the lack of vitamin A, and atrophy of muscle increases because of the unavailability of vitamin E.
After a growth period of human metabolism, there sets in a gradual decline: slow structural changes other than those due to preventable diseases or accidents occur. Aging eventually is characterized by marked atrophy of many tissues and organs, with both a decline in the number of cells and an alteration in their constitution. This is reflected eventually in the changed, diminished, or lost function characteristic of old age and eventuates in death. The changes in senescence are affected by both inherited constitution and environmental influences, including disease and accident.
Atrophic changes of aging affect almost all tissues and organs, but some changes are more obvious and important. Arteriosclerosis—the thickening and hardening of arterial walls—decreases the vascular supply and usually accentuates aging processes.
Atrophy in old age is especially noticeable in the skin, characteristically flat, glossy or satiny, and wrinkled. The atrophy is caused by aging changes in the fibres of the true skin, or dermis, and in the cells and sweat glands of the outer skin. Wasting of muscle accompanied by some loss of muscular strength and agility is common in the aged. In a somewhat irregular pattern, there is shrinkage of many individual muscle fibres as well as a decrease in their number. Other changes have been observed within the muscle cells.
Increase of the pigment lipofuscin is also characteristic in the muscle fibres of the heart in the aged in a condition known as brown atrophy of the heart. Wasting of the heart muscle in old age may be accompanied by increase of fibrous and fatty tissue in the walls of the right side of the heart and by increased replacement of elastic tissue with fibrous tissue in the lining and walls of coronary arteries within the heart muscle. Abnormal deposits of the protein substance amyloid also occur with greater frequency in the atrophic heart muscle in old age.
Atrophy of the liver in the aged is also accompanied by increased lipochrome pigment in the atrophied cells.
The bones become progressively lighter and more porous with aging, a process known as osteoporosis. The reduction of bone tissue is most marked in cancellous bone—the open-textured tissue in the ends of the long bones—and in the inner parts of the cortex of these bones. In addition to changes in and loss of osteocytes, or bone cells, there is decreasing mineralization, or calcium deposit, with enhanced fragility of the bones.
Atrophy of the brain in old age is shown by narrowing of the ridges, or gyri, on the surface of the brain and by increased fluid in the space beneath the arachnoid membrane, the middle layer of the brain covering. There is shrinkage of individual neurons, with an increase in their lipochrome pigment content, as well as a decrease in their number. Sometimes the nerve fibrils have degenerated, and deposits called senile plaques may be found between the neurons, particularly in the frontal cortex and hippocampus (a ridge in the wall of an extension, or horn, of the lateral ventricle, or cavity, of the brain). Similar atrophic changes are seen in the brain in Alzheimer disease, a condition of unknown cause most likely to occur in older patients. The mental deterioration (senile dementia) of the aged is the clinical manifestation of these changes. Senile atrophy may be increased and complicated by the presence of arteriosclerosis.
Simmonds disease is a chronic deficiency of function of the pituitary gland, a form of hypopituitarism, that leads to atrophy of many of the viscera, including the heart, liver, spleen, kidneys, thyroid, adrenals, and gonads. The disease results in emaciation and death if left untreated.
A destructive or atrophic lesion affecting the pituitary gland with loss of hormones leads to atrophy of the thyroid gland, adrenal glands, and gonads and in turn brings atrophic changes to their target organs and the viscera. The decrease in size of the endocrine glands may be extreme.
Atrophy of muscle or of muscle and bone
Local atrophy of muscle, bone, or other tissues results from disuse or diminished activity or function. Although the exact mechanisms are not completely understood, decreased blood supply and diminished nutrition occur in inactive tissues. Disuse of muscle resulting from loss of motor nerve supply to the muscle (e.g., as a result of polio) leads to extreme inactivity and corresponding atrophy. Muscles become limp and paralyzed if there is destruction of the nerve cells in the spinal cord that normally activate them. The shrinkage of the paralyzed muscle fibres becomes evident within a few weeks. After some months, fragmentation and disappearance of the muscle fibres occurs with some replacement by fat cells and a loose network of connective tissue. Some contracture may result.
The skeletal muscles forced to inactivity by paralysis (e.g., of a limb as a result of polio) also undergo disuse atrophy. If there is a tendency for bone to become lighter and more porous in some particular area, a condition known as local osteoporosis, this can be recognized by X-rays within a few weeks. The cortex of the long bones becomes considerably thinned or atrophic, with decreased mineral content. Disuse as a result of painfully diseased joints, as in rheumatoid arthritis, results in a similar but lesser degree of atrophy of muscles concerned with movement of the involved joint, and local atrophy may also occur in the bone in the neighbourhood of the joint. A local osteoporosis of bone known as Sudeck atrophy sometimes develops rapidly in the area of an injury to bone.
Severe or prolonged deficits of blood sugar deprive the nervous system of needed sources of energy and as a rare event result in degeneration of cells of the brain and peripheral nerves. The disuse atrophy of muscle or bone that may result is fundamentally similar to the other disuse atrophies of these tissues.
Persistent pressure will cause atrophy of a compressed cell, organ, or tissue, presumably because of interference with the nutrition and metabolic activity of the affected part. Cells in a local area (e.g., in the liver) atrophy from the pressure of materials such as amyloid deposited around them. The pressure of an expanding benign tumour causes atrophy of adjacent normal structures. The pressure of a localized dilatation of an artery (aneurysm) will cause atrophy of tissues, even bone, on which it impinges.
Bulging of an intervertebral disk or growth of a tumour sometimes brings pressure on nerves near their point of exit from the spinal cord; if the pressure is prolonged, the muscles normally controlled by these nerves may atrophy. Most often the calf muscles are affected. Pressure as a result of involvement of the vertebrae at the level of the neck, or from compression of the network of nerves called the brachial plexus by the scalenus anticus muscle, produces similar effects in the upper chest and arms.
Simple disuse of muscle or bone, as, for example, from the immobilization produced when a limb is put in a cast or sling, results in atrophy of these tissues. In the case of muscle, the degree of atrophy is generally less severe than that caused by injury to a nerve, although the nature of the change is similar.
Localized atrophies of leg and arm muscles may result from hereditary or familial diseases in which the nerves of the spinal cord that supply them are inactivated or destroyed. In Charcot-Marie-Tooth disease, the atrophy involves mainly the peroneal muscles, at the outer side of the lower legs, and sometimes the muscles of the hand as well. It commonly begins in childhood or adolescence. Peroneal muscle atrophy is also seen in the hereditary spinal cord degenerative disease known as Friedreich ataxia.
Atrophy of nerve tissue
Atrophy of brain or spinal cord tissue may be brought about by injuries that directly affect a localized area or that interfere with the blood supply to an area. When peripheral nerves are severed, degenerative and eventually atrophic changes ensue in the part beyond the injury. This type of atrophy is known as Wallerian degeneration. If conditions do not allow regeneration of nerve fibres from the proximal fragment of the cut nerve, atrophy is the eventual fate of the nerve tissue distal to the injury. Retrograde atrophy also occurs from disuse and affects the ganglion cells of the injured nerve.
Prolonged pressure brings about atrophy in the central nervous system as elsewhere. The pressure of an expanding tumour of the membranes covering the brain results in localized atrophy of the adjacent brain substance on which it impinges. In hydrocephalus more widespread atrophy of brain tissue results from the abnormal amounts of fluid confined within the rigid bony compartment of the skull. Increased pressure within the skull may force a portion of the brain through the foramen magnum, the bony opening at the base of the skull, and, if prolonged, results in a localized atrophy of cerebellar tissue pressed against the bony wall.
The late stages of chronic infections may be characterized by atrophy of the brain. A striking example of this is the variety of syphilitic infection of the nervous system known as general paresis in which the brain is shrunk and reduced in weight, the atrophy affecting mainly the cortex of the brain, particularly or most markedly in the frontal area. Occasionally the atrophy is local or affects only one side of the brain. The shrinkage of the brain tissue is mainly due to loss of many nerve cells of the cortex.
Atrophy of fatty tissue
Atrophy of adipose tissue of the body occurs as a part of the generalized atrophy of prolonged undernutrition. Localized atrophy of adipose tissue—lipodystrophy—may be the result of injury to the local area; e.g., repeated insulin injections cause atrophy of fatty tissue at the site of the injections. Progressive lipodystrophy is a disease of unknown cause in which the fatty tissue atrophies only in certain regions of the body. It occurs mainly in women and often begins in childhood. The progressive wasting of adipose tissue affects mainly the face, arms, and trunk. In the affected areas, the specialized fat-holding cells of adipose tissue disappear.
Atrophy of skin
A widespread atrophic change in the skin has been noted as a prominent part of the aging process. Similar atrophic changes in the skin appear to be brought about or enhanced by excessive exposure to sunlight. While a number of abnormal conditions of the skin may include localized atrophic changes in the epidermis or dermis as a part of their lesions, certain generalized diseases of the skin are particularly characterized by such changes. The hardening of the skin known as scleroderma may occur in a localized, or circumscribed, form called morphea or as a more diffuse and severe disease. Advanced stages of scleroderma are characterized by marked atrophy of the tissue and appendages of the true skin. Atrophic thinning of the overlying epidermis also may occur, and the underlying fatty tissue and muscle may atrophy as well. The chronic form of the disease discoid lupus erythematosus also is characterized by atrophy. In advanced stages atrophy occurs particularly in the epidermis in focal areas. The thinned layer of epidermis may be a prominent feature of the microscopic appearance of the skin.
Atrophy of glands
Endocrine glandular tissues may undergo atrophy when an excess of their hormonal product is present as a result of disease. An example is seen in connection with a hormone-producing tumour of the cortical tissue of one adrenal gland, which may be accompanied by marked atrophy of the cortical tissue of the opposite adrenal gland. This probably results from disturbance of the delicate mechanism of hormonal stimulation via the pituitary gland.
Various endocrine organs (thyroid gland, adrenal glands, gonads) depend for their activity on endocrine stimulation by hormones of the pituitary gland. A severe general failure of production of the pituitary hormones results in the widespread endocrine atrophy of Simmonds disease, as has been noted. Lesser degrees of pituitary functional disturbance may disturb a delicate balance, involving mainly one type of stimulating hormone of the pituitary, and may result in selective atrophy of the adrenal cortical tissue or of the gonads.
Glands that release their secretions through a duct (e.g., salivary glands, pancreas) may become atrophic as a result of obstruction of the duct. In the pancreas, a complete obstruction of its duct results in atrophy of the glandular tissue, except for the insulin-producing islets of Langerhans, the secretion of which is absorbed into the bloodstream. Factors of both disuse and increased pressure may be present in the atrophy resulting from obstruction of the outlet channel. Similarly, rapid and complete obstruction of a ureter is followed by atrophy of the corresponding kidney.
Chemical-induced atrophy
Cases of atrophy resulting from chemical injury are not common. In chronic math poisoning, however, degenerative changes occur in peripheral nerves, resulting in weakness and atrophy in the tissues (usually legs or arms) to which the nerves are distributed. Similar results may follow the peripheral neuropathy of chronic lead poisoning.
#282 Re: Dark Discussions at Cafe Infinity » crème de la crème » 2026-02-26 00:21:04
2442) Werner Forssmann
Gist:
Life
Werner Forssmann was born in Berlin, where he also studied medicine. As a newly educated doctor, he served in Eberswalde and conducted his Nobel Prize-awarded experiment there in 1929. His experimentation met resistance, however, which impeded continued research in the field. After being chief surgeon in Dresden and Berlin, Forssmann served as a doctor in the army during World War II. After the war ended, he worked as a district medical officer, among other things. Forssmann and his wife, also a doctor, had six children.
Work
In 1929 the physician Werner Forssmann saw a picture in a book showing how a tube was inserted into the heart of a horse through a vein. A balloon at the other end of the tube showed changes in pressure. Forssmann was convinced that a similar experiment could be carried out on people. Despite the fact that his boss forbade him, Forssmann conducted the experiment on himself. From the crook of his arm he inserted a thin catheter through a vein into his heart and took an X-ray photo. The experiment paved the way for many types of heart studies.
Summary
Werner Forssmann (born Aug. 20, 1904, Berlin, Ger.—died June 1, 1979, Schopfheim, W. Ger.) was a German surgeon who shared with André F. Cournand and Dickinson W. Richards the Nobel Prize for Physiology or Medicine in 1956. A pioneer in heart research, Forssmann contributed to the development of cardiac catheterization, a procedure in which a tube is inserted into a vein at the elbow and passed through the vein into the heart. While a surgical resident in Berlin (1929), Forssmann used himself as the first human subject, watching the progress of the catheter in a mirror held in front of a fluoroscope screen. Forssmann’s daring experiment was condemned at the time as foolhardy and dangerous, and in the face of severe criticism he abandoned cardiology for urology.
Forssmann’s procedure, with slight modifications, was put into practice in 1941 by Richards and Cournand, and has since become an extremely valuable tool in diagnosis and research. It has made possible, among other things, precise measurement of intracardiac pressure and blood flow, injection into the heart of drugs and of opaque material visible on X-ray photographs, and insertion of electrodes for the regulation of the heartbeat.
Forssmann graduated in medicine from the University of Berlin (1928) and then did postgraduate study in urology at Berlin and Mainz. He served as chief of surgery at the city hospital in Dresden-Friedrichstadt and in 1958 was named chief of the surgical division of the Evangelical Hospital in Düsseldorf.
Details
Werner Theodor Otto Forßmann (29 August 1904 – 1 June 1979) was a German researcher and physician from Germany who shared the 1956 Nobel Prize in Medicine (with Andre Frederic Cournand and Dickinson W. Richards) for developing a procedure that allowed cardiac catheterization. In 1929, he put himself under local anesthesia and inserted a catheter into a vein of his arm. Not knowing if the catheter might pierce a vein, he put his life at risk. Forssmann was nevertheless successful; he safely passed the catheter into his heart.
Early life
Forssmann was born in Berlin on 29 August 1904. Upon graduating from Askanisches Gymnasium [de], he entered the University of Berlin to study medicine, passing the State Examination in 1929.
Career
He hypothesized that a catheter could be inserted directly into the heart, for such applications as directly delivering drugs, injecting radiopaque dyes, or measuring blood pressure. The fear at the time was that such an intrusion into the heart would be fatal. To prove his point, he decided to try the experiment on himself.
In 1929, while working in Eberswalde, he performed the first human cardiac catheterization. He ignored his department chief and persuaded the operating-room nurse in charge of the sterile supplies, Gerda Ditzen, to assist him. She agreed, but only on the promise that he would do it on her rather than on himself. However, Forssmann tricked her by restraining her to the operating table and pretending to locally anaesthetise and cut her arm whilst actually doing it on himself. He anesthetized his own lower arm in the cubital region and inserted a urinary catheter into his antecubital vein, threading it partly along before releasing Ditzen (who at this point realised the catheter was not in her arm) and telling her to call the X-ray department. They walked some distance to the X-ray department on the floor below where under the guidance of a fluoroscope he advanced the catheter the full 60 cm into his right ventricular cavity. This was then recorded on X-ray film showing the catheter lying in his right atrium.
The head clinician at Eberswalde, although initially very annoyed, recognized Werner's discovery when shown the X-rays; he allowed Forssmann to carry out another catheterization on a terminally ill woman whose condition improved after being given drugs in this way. An unpaid position was created for Forssmann at the Berliner Charité Hospital, working under Ferdinand Sauerbruch, although once Sauerbruch saw his paper, he was dismissed for continuing without his approval. Sauerbruch commented, "You certainly can't begin surgery in that manner". Facing such disciplinary action for self-experimentation, he was initially forced to leave the Charité, but was later reinstated until again being forced to leave in 1932 for not meeting scientific expectations. His surgical skills were noted, however, and he was recommended to another hospital where he worked for a while before leaving in 1933 after marrying Dr. Elsbet Engel, a specialist in urology there. Finding it difficult to get a job with his reputation, he quit cardiology and took up urology. He then went on to study urology under Karl Heusch at the Rudolf Virchow Hospital [de] in Berlin. Later, he was appointed chief of the surgical clinic at both the City Hospital at Dresden-Friedrichstadt and the Robert Koch Hospital [de] in Berlin.
From 1932 to 1945, he was a member of the Nazi Party. At the start of World War II, he became a medical officer. In the course of his service, he rose to the rank of major, until he was captured and put into a U.S. POW camp. Upon his release in 1945, he worked as a lumberjack and then as a country medic in the Black Forest with his wife. In 1950, he began practice as a urologist in Bad Kreuznach.
During the time of his imprisonment, his paper was read by André Frédéric Cournand and Dickinson W. Richards. They developed ways of applying his technique to heart disease diagnosis and research. In 1954, he was given the Leibniz Medal of the German Academy of Sciences. In 1956, the Nobel Prize in Physiology or Medicine was awarded to Cournand, Richards, and Forßmann.
After winning the Nobel Prize, he was given the position of honorary professor of surgery and urology at the University of Mainz. In 1961, he became an honorary professor at the National University of Córdoba. In 1962, he became a member of the executive board of the German Society of Surgery. He also became a member of the American College of Chest Physicians, honorary member of the Swedish Society of Cardiology, the German Society of Urology [de], and the German Child Welfare Association.
Personal life
He and Elsbet had six children: Klaus Forßmann in 1934, Knut Forßmann in 1936, Jörg Forßmann in 1938, Wolf Forßmann in 1939 (who was first to isolate the atrial natriuretic peptide), Bernd Forßmann in 1940 (who helped develop the first clinical lithotriptor), and Renate Forßmann in 1943.
He died in Schopfheim, Germany, of heart failure on 1 June 1979. His wife died in 1993.

#283 Jokes » Junk Food Jokes - III » 2026-02-26 00:06:27
- Jai Ganesh
- Replies: 0
Q: What does it do before it rains candy?
A: It sprinkles!
* * *
Q: When should you take a cookie to the doctor?
A: When it feels crummy.
* * *
Q: What does a gambling addict eat?
A: Poker chips and salsa.
* * *
Q: What is a monkey's favorite cookie?
A: Chocolate chimp!
* * *
Q: What does an excited fat kid do in the junk food isle?
A: The Moon-Pies Walk.
* * *
#284 Dark Discussions at Cafe Infinity » Come Quotes - XIV » 2026-02-26 00:06:02
- Jai Ganesh
- Replies: 0
Come Quotes - XIV
1. It is important that those that do come to settle in the U.K. gain a sense of British identity and share British values. We have always attracted the best and brightest. - Rishi Sunak
2. The time has come for us to draw the line. The time has come for the responsible leaders of both political parties to take a stand against overgrown Government and for the American taxpayer. - Richard M. Nixon
3. Now, as a nation, we don't promise equal outcomes, but we were founded on the idea everybody should have an equal opportunity to succeed. No matter who you are, what you look like, where you come from, you can make it. That's an essential promise of America. Where you start should not determine where you end up. - Barack Obama
4. Every beauty which is seen here by persons of perception resembles more than anything else that celestial source from which we all are come. - Michelangelo
5. The tennis ball doesn't know how old I am. The ball doesn't know if I'm a man or a woman or if I come from a communist country or not. Sport has always broken down these barriers. - Martina Navratilova
6. The scientific man does not aim at an immediate result. He does not expect that his advanced ideas will be readily taken up. His work is like that of the planter - for the future. His duty is to lay the foundation for those who are to come, and point the way. - Nikola Tesla
7. Clouds come floating into my life, no longer to carry rain or usher storm, but to add color to my sunset sky. - Rabindranath Tagore
8. I have come to the conclusion that politics are too serious a matter to be left to the politicians. - Charles de Gaulle.
#285 This is Cool » Motor Neuron » 2026-02-25 18:13:13
- Jai Ganesh
- Replies: 0
Motor Neuron
Gist
Motor neurons (or motoneurons) are specialized efferent nerve cells originating in the brain or spinal cord that transmit impulses to effector muscles and glands, controlling voluntary and involuntary movements. They are classified into upper motor neurons (brain to spinal cord) and lower motor neurons (spinal cord to muscle).
Motor neurones are cells in the brain and spinal cord that allow us to move, speak, swallow and breathe by sending commands from the brain to the muscles that carry out these functions.
Summary
Motor neurons, also known as efferent neurons, are nerve cells responsible for carrying central nervous system signals towards muscles to cause voluntary or involuntary movement through the innervation of effector muscles and glands. Their nerve fibers are considered to be the longest in the human body. Motor neurons are the most common structure for neurons.
Motor neurons are divided into either upper or lower motor neurons. Each type of motor neuron utilizes different neurotransmitters to relay their signals.
* Upper motor neurons originate primarily in the cerebral cortex (primary motor cortex) and extend towards the brainstem or spinal cord, where they synapse. Upper motor neurons travel down specific pathways, including the pyramidal, extrapyramidal, rubrospinal, tectospinal and reticulospinal tracts. Glutamate is used as a neurotransmitter.
* Lower motor neurons originate in the brainstem (cranial nerve nuclei) and the spinal cord (anterior horn) and project to innervate muscles and glands throughout the body. Acetylcholine is used as a neurotransmitter.
The structure of a motor neuron is characterized by three components: the soma, the axon, and the dendrites. Motor neurons have a large cell body, or soma, and long projections used in transmitting information away from the soma. These projections are referred to as axons and dendrites. Axons send impulses away from the soma and dendrites carry incoming information. Motor neurons are typically considered multipolar in terms of their structure, which translates to having a single axon and multiple dendrites.
Myelin sheath surrounds the axon of the motor neuron. One of the key functions of myelin is to insulate the axon length ensuring that electrical impulses are transmitted quickly and efficiently to the target structure. Each motor neuron divides into many terminal branches which synapse with muscle fibers at neuromuscular junctions (NMJ), also known as myoneural junctions. Electrical impulses are transmitted from the motor neuron to the muscle fiber via axon terminals (also known as neuropodia or axon endfeet), to generate muscle contractions. Axon terminals function to release neurotransmitters which are received by specific parts of the muscle fiber membrane known as motor end plates, signalling muscle contraction.
A motor neuron and its associated muscle fibers make up a motor unit. Fine muscles (e.g. extraocular muscles) have small motor units and therefore can be controlled more precisely in comparison to larger muscles (e.g. biceps brachii).
Details
A motor neuron (or motoneuron), also known as efferent neuron is a neuron that allows for both voluntary and involuntary movements of the body through muscles and glands. Its cell body is located in the motor cortex, brainstem or the spinal cord, and whose axon (fiber) projects to the spinal cord or outside of the spinal cord to directly or indirectly control effector organs, mainly muscles and glands. There are two types of motor neuron – upper motor neurons and lower motor neurons. Axons from upper motor neurons synapse onto interneurons in the spinal cord and occasionally directly onto lower motor neurons. The axons from the lower motor neurons are efferent nerve fibers that carry signals from the spinal cord to the effectors. Types of lower motor neurons are alpha motor neurons, beta motor neurons, and gamma motor neurons.
A single motor neuron may innervate many muscle fibres and a muscle fibre can undergo many action potentials in the time taken for a single muscle twitch. Innervation takes place at a neuromuscular junction and twitches can become superimposed as a result of summation or a tetanic contraction. Individual twitches can become indistinguishable, and tension rises smoothly eventually reaching a plateau.
Although the word "motor neuron" suggests that there is a single kind of neuron that controls movement, this is not the case. Indeed, upper and lower motor neurons—which differ greatly in their origins, synapse locations, routes, neurotransmitters, and lesion characteristics—are included in the same classification as "motor neurons." Essentially, motor neurons, also known as motoneurons, are made up of a variety of intricate, finely tuned circuits found throughout the body that innervate effector muscles and glands to enable both voluntary and involuntary motions. Two motor neurons come together to form a two-neuron circuit. While lower motor neurons start in the spinal cord and go to innervate muscles and glands all throughout the body, upper motor neurons originate in the cerebral cortex and travel to the brain stem or spinal cord. It is essential to comprehend the distinctions between upper and lower motor neurons as well as the routes they follow in order to effectively detect these neuronal injuries and localise the lesions.
Disorders causing selective injury to motor neurons are classified as motor neuron diseases.
Additional Information
Motor neurons are nerve cells that carry messages from the brain and spinal cord to your muscles and glands. They control everything from blinking and walking to breathing and digestion.
By acting as messengers between the central nervous system (CNS) and the body, they make movement and essential bodily functions possible.
Motor neurons belong to a broader group called efferent neurons—cells that send signals outward from the CNS. These neurons are crucial for both voluntary actions, like typing, and involuntary ones, like maintaining your posture.
Key Takeaways
* Motor neurons carry messages from your brain and spinal cord to your muscles.
* They’re essential for movement, reflexes, and basic body functions.
* There are two main types: upper and lower motor neurons.
* Damage can cause serious issues, including paralysis and diseases like ALS.
* While some treatments exist, prevention and early detection are key.
(ALS: Amyotrophic lateral sclerosis)
How Do Motor Neurons Work?
Motor neurons act like messengers. They carry electrical signals from your brain or spinal cord to your muscles.
When the message reaches its destination, it triggers the release of a chemical called acetylcholine at a point where the nerve meets the muscle—this spot is called the neuromuscular junction.
Acetylcholine tells your muscle to contract, which produces movement.
Motor neurons have three main parts:
* Soma (cell body): The cell’s headquarters, where energy and proteins are produced.
* Dendrites: Short branches that receive messages from other neurons.
* Axon: A long fiber that carries the outgoing message to a muscle or gland.
Each part works together to ensure your body reacts smoothly and quickly.
What Are the Types of Motor Neurons?
Motor neurons are divided into two main groups, based on where they begin and what they control:
Upper Motor Neurons
These begin in the motor cortex of the brain or the brainstem. They send signals down to the spinal cord to help start and guide movement.
Key pathways include:
* Pyramidal tract: Controls deliberate, voluntary actions like lifting your arm.
* Extrapyramidal tracts: Manage automatic functions like balance and posture. This includes:
* Rubrospinal tract: Helps adjust body balance.
* Tectospinal tract: Affects neck muscle movement.
* Reticulospinal tract: Regulates automatic actions.
Lower Motor Neurons
These are the final link between your nervous system and your muscles. They carry messages directly to muscle fibers.
Lower motor neurons are categorized into:
* Somatic Motor Neurons:
** Alpha neurons: Connect to standard muscle fibers to produce forceful contractions.
** Beta neurons: Interact with both standard and sensory muscle fibers.
** Gamma neurons: Fine-tune muscle tone by connecting to stretch sensors.
* Special Visceral Efferent Neurons: Found in the brainstem, these are part of cranial nerves like the facial nerve and vagus nerve, which control facial expressions and digestion.
* General Visceral Motor Neurons: Part of the autonomic nervous system, they connect to organs like the heart and intestines.
What Do Motor Neurons Do?
Motor neurons allow your brain to control your body. Their responsibilities include:
* Voluntary movements: Walking, writing, smiling
* Involuntary reflexes: Pulling your hand away from something hot
* Autonomic functions: Breathing, heartbeat, digestion
They’re also essential for posture, balance, and muscle coordination—things we often take for granted.
Location
Motor neurons are located in the central nervous system (CNS), specifically in the motor cortex, brainstem, and spinal cord.
While the cell bodies (soma) of motor neurons remain within the CNS, their axons, called efferent fibers, project outward to reach muscles and other peripheral systems such as organs and glands.
These neurons are notable for having some of the longest axons in the body. In fact, the efferent fibers that extend from the base of the spinal cord to the toes represent one of the longest axon pathways in the human body.
With approximately 500,000 motor neurons in total, these cells form an extensive network carrying information from the CNS to peripheral organs, muscles, and glands.

#286 Re: Jai Ganesh's Puzzles » General Quiz » 2026-02-25 17:38:45
Hi,
#10767. What does the term in Biology Guanine mean?
#10768. What does the term in Biology Habitat mean?
#287 Re: Jai Ganesh's Puzzles » English language puzzles » 2026-02-25 17:29:58
Hi,
#5963. What does the noun nectar mean?
#5964. What does the noun nectarine mean?
#288 Re: Jai Ganesh's Puzzles » Doc, Doc! » 2026-02-25 17:14:55
Hi,
#2578. What does the medical term Chlorambucil mean?
#289 Science HQ » Specific Heat » 2026-02-25 16:56:56
- Jai Ganesh
- Replies: 0
Specific Heat
Gist
In thermodynamics, the specific heat capacity (symbol c) of a substance is the amount of heat that must be added to one unit of mass of the substance in order to cause an increase of one unit in temperature. It is also referred to as massic heat capacity or as the specific heat.
Specific heat (or specific heat capacity) is the amount of heat energy needed to raise the temperature of one unit of mass (like one gram or kilogram) of a substance by one degree Celsius (or Kelvin). It's a measure of how much heat a material can store, often called its thermal inertia, and substances with high specific heat, like water, resist temperature changes more than those with low specific heat, like gold. This property is crucial in understanding everything from cooking to climate science.
Summary
The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius. The relationship between heat and temperature change is usually expressed in the form shown below where c is the specific heat. The relationship does not apply if a phase change is encountered, because the heat added or removed during a phase change does not change the temperature.
The specific heat of water is 1 calorie/gram °C = 4.186 joule/gram °C which is higher than any other common substance. As a result, water plays a very important role in temperature regulation. The specific heat per gram for water is much higher than that for a metal, as described in the water-metal example. For most purposes, it is more meaningful to compare the molar specific heats of substances.
The molar specific heats of most solids at room temperature and above are nearly constant, in agreement with the Law of Dulong and Petit. At lower temperatures the specific heats drop as quantum processes become significant. The low temperature behavior is described by the Einstein-Debye model of specific heat.
The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius. The relationship between heat and temperature change is usually expressed in the form shown below where c is the specific heat. The relationship does not apply if a phase change is encountered, because the heat added or removed during a phase change does not change the temperature.
Details:
Understanding Specific Heat Capacity
This concept is fundamental to thermodynamics and heat transfer, since it expresses the amount of heat energy that must be supplied to a unit of mass of a substance to raise its temperature by one degree Celsius. This concept has several important applications in practice. Engineers and materials scientists use it to choose the right materials that will bring rapid temperature changes where needed and ensure thermal stability where necessary.
What is Specific Heat?
It is the amount of heat energy required to raise any substance’s temperature by one degree Celsius. This is an intrinsic property of the material and depends upon the nature of the substance. It is one of the most critical attributes for scientists and engineers because it touches areas from climate science to culinary arts. 1
Understanding this characteristic means that different materials, upon receiving the same amount of heat, change temperature by different amounts. This is due to the molecular structure and bonding nature of each substance. Imagine you are at the beach on a hot day. The sand feels extremely hot, while the water does not. It is because sand has a low specific heat, it warms relatively quickly. On the other hand, water’s high specific heat allows it to absorb more heat without raising its temperature as quickly.
What is Heat Capacity?
It is defined as the amount of heat energy required to increase the entire object’s temperature by one degree Celsius. Heat capacity does depend upon the mass of an object and its composition.
For example, while specific heat for both a large block of iron and a small iron nail is the same; heat capacity will be different as the large block of iron will need more heat to raise the temperature by one degree while the small iron nail would need relatively less heat to achieve the same temperature increase.
Some Examples of Specific Heat Capacity
Water (liquid) 4.18 J/kg/C: Water has great specific capacity, which is one of its best qualities since it helps it to conduct heat effectively. It can absorb and release massive amounts of energy without undergoing much variation in temperatures. For instance, car radiator systems and electricity generation amid other industries. 4
Steam 2.01 J/kg/C: The specific heat of steam is important in the design of steam engines and turbines. Since engineers must know how much energy is needed to make steam, and they also must know how much heat can be carried by the steam, they shall view power generation systems in relation to their efficiency.
Copper 0.385J/kg/C: The specific heat capacity of copper applies to both electrical engineers and plumbers because it can efficiently conduct heat. This property is very important when creating any electrical wiring or electronic circuit boards that involve heat exchange. This ensures the proper management of temperatures within an electronic device and system.
Iron 0.449J/kg/C: Iron’s specific heat capacity is an important factor not only in the manufacturing of cookware like pans and skillets but also in the construction industry. This property allows iron to effectively absorb and retain heat, making it valuable for creating materials that require durability and thermal stability.
Key Takeaways
For many scientific and engineering applications, it is important to understand specific heat capacity and its relationship with regards to heat capacity. The main messages that could be drawn from this discussion are below.
* Specific heat capacity: The amount of heat that needs to be supplied in order to raise the unit mass of substance by 1 degree Celsius.
* Heat Capacity vs Specific Heat Capacity: Heat capacity is dependent on the mass and composition of the object, but specific heat capacity is an intrinsic attribute of a material.
* In the case of gases, the specific heat capacity may be different while the process is occurring at constant pressure or at constant volume.
* In the case of solids and liquids, the difference between specific heat capacity at constant pressure and at constant volume may be considered negligible.
Therefore, it is a fundamental concept that connects theoretical understanding to practical applications. It plays a critical role in everything from industrial machinery to household appliances, enabling informed decisions about performance, safety, and energy efficiency. To ensure you’re making the best choices, it’s vital to have accurate instruments to measure thermal properties. Explore Thermtest’s product line to find the right tools for your needs.
Frequently Asked Questions:
What does the specific heat depend on?
Specific heat depends on the material’s molecular structure and bonding. Substances with stronger molecular bonds typically have higher specific heat because more energy is required to increase their temperature.
What is the specific heat of water?
The specific heat capacity of water at room temperature and pressure is approximately 4.18 J/g°C.
What material has the highest specific heat capacity?
Water has one of the highest specific heat capacities among common substances, at 4.18 J/g°C. This is because of the hydrogen bonding between water molecules, which requires significant energy to overcome. 5
How do you measure specific heat capacity?
It can be measured using a calorimeter, which quantifies the amount of heat transferred to or from a substance as its temperature changes.
What is the difference between heat capacity and specific heat?
Heat Capacity measures the heat required to raise the substance’s temperature by 1 degree. On the other hand, specific heat capacity is the amount of heat required to raise the temperature of 1 kg of a substance by 1 degree. While the former depends on the total mass or the amount of substance, the later does not. That is why the concept of heat capacity is used to understand how much an object can absorb or release heat during a given temperature change, and the concept of specific heat capacity is helpful in relating thermal properties of different materials.
What is the SI unit of specific heat capacity?
The SI unit is joules per kilogram per Kelvin (J/kg·K).
Additional Information
In thermodynamics, the specific heat capacity (symbol c) of a substance is the amount of heat that must be added to one unit of mass of the substance in order to cause an increase of one unit in temperature. It is also referred to as massic heat capacity or as the specific heat. More formally it is the heat capacity of a sample of the substance divided by the mass of the sample.
Specific heat capacity often varies with temperature, and is different for each state of matter.
While the substance is undergoing a phase transition, such as melting or boiling, its specific heat capacity is technically undefined, because the heat goes into changing its state rather than raising its temperature.
Specific heat is the quantity of heat required to raise the temperature of one gram of a substance by one Celsius degree. The units of specific heat are usually calories or joules per gram per Celsius degree. For example, the specific heat of water is 1 calorie (or 4.186 joules) per gram per Celsius degree. The Scottish scientist Joseph Black, in the 18th century, noticed that equal masses of different substances needed different amounts of heat to raise them through the same temperature interval, and, from this observation, he founded the concept of specific heat. In the early 19th century the French physicists Pierre-Louis Dulong and Alexis-Thérèse Petit demonstrated that measurements of specific heats of substances allow calculation of their atomic weights

#290 Re: Jai Ganesh's Puzzles » 10 second questions » 2026-02-25 14:53:15
Hi,
#9864.
#291 Re: Jai Ganesh's Puzzles » Oral puzzles » 2026-02-25 14:39:54
Hi,
#6358.
#292 Re: Exercises » Compute the solution: » 2026-02-25 14:25:47
Hi,
2718.
#293 Re: This is Cool » Miscellany » 2026-02-25 00:48:00
2504) Hydraulic Press
Gist
A hydraulic press works on the principle of Pascal's law, which states that when pressure is applied to a confined fluid, the pressure change occurs throughout the entire fluid.
In hydraulic presses, Pascal's law is applied: pressure in a confined fluid is transmitted equally in all directions. In mechanical presses, kinetic energy is stored in a flywheel and transmitted via clutch/crank mechanisms to the ram.
Summary
A hydraulic press uses a hydraulic cylinder to produce a compressive force. Within a hydraulic press, there is a plate where the sample is placed to be pressed for sample preparation.
How Does a Hydraulic Press Work?
A hydraulic press works on the principle of Pascal’s law, which states that when pressure is applied to a confined fluid, the pressure change occurs throughout the entire fluid. Within the hydraulic press, there is a piston that works as a pump, that provides a modest mechanical force to a small area of the sample. There is also a piston with a larger area, which produces a larger mechanical force.
Advantages
There are a variety of advantages to using hydraulic presses. This is evident in hand-fed hydraulic systems, where the ease and speed of sample switching is considerably improved.
These instruments are also extremely beneficial for high-volume sample preparation, also increasing the speed of sampling.
Details
A hydraulic press is a machine press using a hydraulic cylinder to generate a compressive force. It uses the hydraulic equivalent of a mechanical lever, and was also known as a Bramah press after the inventor, Joseph Bramah. He invented and was issued a patent on this press in 1795. As Bramah installed toilets and developed the modern flush toilet, he studied existing literature on the motion of fluids to develop the press.
Main principle
The hydraulic press depends on Pascal's principle. The pressure throughout a closed system is constant. One part of the system is a piston acting as a pump, with a modest mechanical force acting on a small cross-sectional area; the other part is a piston with a larger area which generates a correspondingly large mechanical force. Only small-diameter tubing (which more easily resists pressure) is needed if the pump is separated from the press cylinder.
Application
Hydraulic presses are commonly used for assembly and disassembly of tightly-fitting components. In manufacturing, they are used for forging, clinching, molding, blanking, punching, deep drawing, and metal forming operations. Hydraulic presses are also used for stretch forming, rubber pad forming, and powder compacting. The hydraulic press is advantageous in manufacturing, as it gives the ability to create more intricate shapes than other methods and can be economical with materials. A hydraulic press will take up less space compared to a mechanical press of the same capability. Hydraulic presses are also used for waste processing operations such as in garbage trucks and car crushers to reduce the size of waste material for easier more economic transportation.
In geology a tungsten carbide coated hydraulic press is used in the rock crushing stage of preparing samples for geochemical analyses in topics such as understanding the origins of volcanism.
In popular culture
The room featured in Fermat's Room has a design similar to that of a hydraulic press. Boris Artzybasheff also created a drawing of a hydraulic press, in which the press was created out of the shape of a robot.
In 2015, the Hydraulic Press Channel, a YouTube channel dedicated to crushing objects with a hydraulic press, was created by Lauri Vuohensilta, a factory owner from Tampere, Finland. The Hydraulic Press Channel has since grown to over 10 million subscribers on YouTube. There are numerous other YouTube channels that publish videos involving hydraulic presses that are tasked with crushing many different items, such as bowling balls, soda cans, plastic toys, and metal tools.
A hydraulic press is featured prominently in the Sherlock Holmes story "The Adventure of the Engineer's Thumb".
Additional Information
A hydraulic press, by definition, is a mechanical apparatus that leverages the static pressure of a liquid as explained by Pascal's principle. It's employed to form, reshape, and alter various materials including metals, plastics, rubber, and wood. The primary components of a hydraulic press comprise the mainframe, the power system, and the control mechanisms.
According to Pascal's principle, if pressure is exerted on a confined fluid, the pressure change spreads consistently throughout the liquid. In a hydraulic press, this applied pressure emanates from a piston, acting similarly to a pump to produce mechanical force.
The hydraulic press is a powerful machine widely used in industrial applications for metal forming, assembly, molding, and material compaction. The hydraulic press operates by forcing hydraulic fluid into a double-acting piston, relying on Pascal's Law to multiply force for heavy-duty tasks. The compressive force generated within the smaller hydraulic cylinder pushes the hydraulic oil (fluid) into a larger cylinder, where much greater force and pressure are applied. As the larger piston moves, it forces the fluid back into the smaller piston cylinder, maintaining a constant hydraulic pressure throughout the system.
This controlled fluid exchange between the two pistons increases mechanical pressure, creating the substantial force needed to drive the anvil of the hydraulic press downward onto a workpiece, shaping, compacting, or cutting the material into its desired form. Once the forming, punching, or pressing process is complete, the pressure is safely released, allowing the anvil and die to return to their original positions. Hydraulic presses are highly valued for their precision, repeatability, and ability to perform a variety of material processing operations, making them a cornerstone in manufacturing, automotive, aerospace, and fabrication industries.
How a Hydraulic Press Works
A typical hydraulic press consists of two cylinders, two pipes, and two pistons. One cylinder functions as the ram, while the other acts as the plunger; they are connected by a high-strength chamber filled with hydraulic fluid, often a specialized oil designed for high-pressure environments. The configuration of these components directly enables the powerful compression and shaping abilities for which hydraulic presses are known.
* Main Cylinder (Ram): Delivers force to the workpiece.
* Plunger: Initiates pressure transfer by pushing hydraulic fluid.
* Pipes and Hoses: Ensure secure fluid movement between components, preventing leaks and ensuring safety.
* Hydraulic Fluid Reservoir: Stores and supplies fluid to support continuous operation.
* Control Valves: Direct fluid flow, manage pressure, and ensure precise operation for various press functions such as forging, stamping, and embossing.
Ram
In advanced hydraulic systems, multiple rams are used, and the number depends on the required working load. Utilizing several smaller rams rather than a single larger one allows for finer control over the hydraulic thrust force, which is essential for delicate metal forming or precision stamping. Hydraulic fluid is routed to these rams by a hydraulic pump and an accumulator, which moderates pressure between the rams and pump, ensuring optimal performance for high-tonnage applications such as deep drawing, blanking, or coining.
Accumulator
The hydraulic accumulator is a critical component that stores pressurized hydraulic fluid, releasing it as needed to maintain system efficiency and responsiveness. It typically consists of a durable cylinder with a spring-loaded or pneumatic piston separator. The accumulator functions as an energy storage device, enabling the hydraulic press to quickly respond to sudden demands without requiring the hydraulic pump to operate continuously. The constant pressure provided by the accumulator is crucial for consistent operation in heavy manufacturing processes, energy efficiency, and reducing mechanical wear. Properly sized accumulators enhance the safety, speed, and reliability of industrial hydraulic press systems.
Pump
There are three main types of hydraulic pumps used in presses: vane, gear, and piston, with piston pumps prevailing in high-performance equipment due to their efficiency and durability under high loads. Hydraulic presses utilize positive displacement pumps, which deliver a precise, constant volume of hydraulic oil during each cycle, maintaining high forces required in automotive component manufacturing, metal stamping, and plastic molding applications. Positive displacement pumps can be either fixed or variable; fixed pumps operate at set speeds, while variable pumps allow for speed and direction changes, enabling customized pressure control based on specific pressing or forming requirements. Piston pumps—available as axial, bent axis, or radial designs—are especially suited for hydraulic press systems because they handle high pressure, provide superior volumetric efficiency, and minimize fluid leakage, ensuring long-term operational reliability in industrial and commercial environments.
Cylinders
The number and size of cylinders in a hydraulic press depend on the design and intended tonnage capacity. These cylinders generate the immense compressive force that drives the press ram and die to shape or cut metal, plastic, rubber, or composites. In a typical two-cylinder design, the cylinder paired with the ram has a larger diameter to produce maximum force, while the plunger cylinder is smaller and initiates fluid movement. Both are made from robust, pressure-rated steel, featuring input/output ports for controlled hydraulic fluid circulation to support heavy pressing operations.
Cylinders are interconnected via hydraulic pipes and hoses, which transmit high-pressure fluid. When the plunger's downward stroke applies pressure to the hydraulic oil, the resulting force is transferred to the main ram. This unidirectional force—delivered via a linear actuator—enables consistent, dynamic motion, critical for applications such as metalforming, compression molding, powder compacting, and precision stamping.
Hydraulic Press Process
The hydraulic press process is the foundation of modern metal forming, fabrication, and materials engineering. The animation below demonstrates how consistent hydraulic pressure from the system's cylinders powers the main ram, driving the press anvil downward onto the workpiece or slug positioned in the die cavity. The result is precise, repeatable compression, shaping, or assembly of materials into components for the automotive, aerospace, electronics, and appliance industries. Because hydraulic presses can deliver adjustable force, stroke length, and speed, they are ideal for a diverse range of processes, including forging, deep drawing, extrusion, lamination, and powder metallurgy. Their versatility and controllability ensure ideal results and cost-effective production in both high-volume and custom-manufacturing environments.
Choosing the Right Hydraulic Press for your production needs involves evaluating not only the type of hydraulic system, but also key factors such as required tonnage, workspace dimensions, safety features, energy consumption, and cycle speed. Leading hydraulic press manufacturers offer custom solutions designed for processes such as metal stamping, plastic injection, rubber molding, and powder compaction. When evaluating suppliers and comparing models, consider the quality of components (cylinders, pumps, seals, frames), after-sales technical support, and maintenance requirements to ensure long-term reliability and maximum return on investment.

#294 Re: Dark Discussions at Cafe Infinity » crème de la crème » 2026-02-25 00:16:38
2441) André Frédéric Cournand
Gist:
Life
André Cournand was born in Paris. His father was a doctor, and Cournand studied natural science and medicine in Paris. After becoming a medical doctor, he made his way to the U.S. in 1930 to spend a year at Bellevue Hospital and Columbia University in New York. There he began a prolonged collaboration with Dickinson Richards and decided to stay in the U.S. Cournand married Sibylle Blumer, who was a widow, and adopted her son. The couple had three more children.
Work
Even though Werner Forssmann succeeded in inserting a catheter into his own heart in 1929, there was great hesitance about continuing this type of research. Nonetheless, beginning in 1941 André Cournand and Dickinson Richards published a series of studies that established use of cardiac catheterization, among other things, to introduce contrast fluid for X-ray images and to measure pressure and oxygen content. Because it was possible to reach the upper chambers of the heart, blood pressure and the blood’s oxygen content could be measured on the way from the heart to the lungs, which was impossible before.
Summary
André F. Cournand (born Sept. 24, 1895, Paris, France—died Feb. 19, 1988, Great Barrington, Mass., U.S.) was a French-American physician and physiologist who in 1956 shared the Nobel Prize for Physiology or Medicine with Dickinson W. Richards and Werner Forssmann for discoveries concerning heart catheterization and circulatory changes.
His medical studies interrupted by World War I, Cournand graduated from the University of Paris in 1930. He studied at Bellevue Hospital, New York City, where he met Richards. Together they collaborated in clinical lung and heart research and perfected Forssmann’s procedure, now termed cardiac catheterization, whereby a tube is passed into the heart from a vein at the elbow. With this procedure it became possible to study the functioning of the diseased human heart and to make more accurate diagnoses of the underlying anatomic defects. Cournand and Richards also used the catheter to examine the pulmonary artery, thus enabling improvement in the diagnosis of lung diseases as well.
Cournand joined the faculty of the College of Physicians and Surgeons of Columbia University in 1934, retiring as emeritus professor of medicine in 1964. He remained active as a special lecturer until his final illness. He became a naturalized citizen of the United States in 1941.
Details
André Frédéric Cournand (September 24, 1895 – February 19, 1988) was a French-American physician and physiologist.
Biography
Cournand was awarded the Nobel Prize in Physiology or Medicine in 1956 along with Werner Forssmann and Dickinson W. Richards for the development of cardiac catheterization.
Born in Paris, Cournand emigrated to the United States in 1930 and, in 1941, became a naturalized citizen. For most of his career, Cournand was a professor at the Columbia University College of Physicians and Surgeons and worked at Bellevue Hospital in New York City.
Many seats of medical research have recognized his work, and he has received the Anders Retzius Silver Medal of the Swedish Society for Internal Medicine (1946), the Albert Lasker Award for Basic Medical Research (1949), the John Philipps Memorial Award of the American College of Physicians (1952), the Gold Medal of the Académie Royale de Médecine de Belgique and of the Académie Nationale de Médecine, Paris (1956). He was elected Doctor (honoris causa) of the Universities of Strasbourg (1957), Lyon (1958), Brussels (1959), Pisa (1961), and D.Sc. of the University of Birmingham (1961).
In 1981, Cournand became a founding member of the World Cultural Council.
His widow Beatrice died in 1993 aged 90.

#295 Jokes » Junk Food Jokes - II » 2026-02-25 00:04:23
- Jai Ganesh
- Replies: 0
Q: What do you get if you divide the circumference of a bowl of ice cream by its diameter?
A: Pi a'la mode.
* * *
Q: Why did the ice cream truck break down?
A: Because of the Rocky Road.
* * *
Q: When can a pizza marry a hot dog?
A: After they have a very frank relationship!
* * *
Q: What is a man's idea of a balanced diet?
A: A bag of potato chips in each hand!
* * *
Q: How do you learn how to make ice cream?
A: In Sunday (Sundae) School.
* * *
#296 Dark Discussions at Cafe Infinity » Come Quotes - XIII » 2026-02-25 00:04:04
- Jai Ganesh
- Replies: 0
Come Quotes - XIII
1. I dream for a living. Once a month the sky falls on my head, I come to, and I see another movie I want to make. - Steven Spielberg
2. True love doesn't come to you it has to be inside you. - Julia Roberts
3. People always fear change. People feared electricity when it was invented, didn't they? People feared coal, they feared gas-powered engines... There will always be ignorance, and ignorance leads to fear. But with time, people will come to accept their silicon masters. - Bill Gates
4. The talent of success is nothing more than doing what you can do well, and doing well whatever you do without thought of fame. If it comes at all it will come because it is deserved, not because it is sought after. - Henry Wadsworth Longfellow
5. I happened to come along in the music business when there was no trend. - Elvis Presley
6. I live now on borrowed time, waiting in the anteroom for the summons that will inevitably come. And then - I go on to the next thing, whatever it is. One doesn't, luckily, have to bother about that. - Agatha Christie
7. All treaties between great states cease to be binding when they come in conflict with the struggle for existence. - Otto von Bismarck
8. Now, as a nation, we don't promise equal outcomes, but we were founded on the idea everybody should have an equal opportunity to succeed. No matter who you are, what you look like, where you come from, you can make it. That's an essential promise of America. Where you start should not determine where you end up. - Barack Obama.
#297 This is Cool » Cyclotron » 2026-02-24 23:30:03
- Jai Ganesh
- Replies: 0
Cyclotron
Gist
A cyclotron is a compact particle accelerator that uses a constant magnetic field to bend charged particles into a spiral path and a rapidly varying electric field to accelerate them to high speeds. Invented by Ernest Lawrence in 1929–1930, it is primarily used in medicine to produce short-lived radioisotopes for cancer diagnosis (PET scans) and treatment. (PET : Positron Emission Tomography).
Cyclotrons are particle accelerators used in medicine (producing radioisotopes for imaging/therapy, cancer treatment via proton beams) and nuclear physics research (bombarding nuclei for experiments, studying atomic properties, creating new elements/isotopes). Their compact size makes them practical for generating high-energy particle beams for these scientific and medical applications, offering advantages over linear accelerators in certain scenarios.
Summary
A cyclotron is a particle accelerator that uses magnetic and electric fields to speed up charged particles to very high speeds and powers many of the tools, treatments, and discoveries that improve our daily lives.
If you have ever had a PET scan at a hospital or heard about radiation treatment for cancer or brain tumours, there's a good chance a cyclotron was involved.
But what is a cyclotron and how is it used?
Let’s break it down.
A cyclotron is a type of particle accelerator. It uses magnetic and electric fields to speed up charged particles like protons or ions to very high speeds. This allows the particles to collide with target materials to produce radioisotopes through nuclear reactions.
Radioisotopes have several uses, including in life saving medical treatments, scientific research, and even clean energy technologies.
The cyclotron was invented in 1931 by American physicist Ernest O. Lawrence and his student M. Stanley Livingston at the University of California, Berkeley. Their early prototype - just about 10 cm wide - was already capable of accelerating particles to high energy levels. Lawrence’s groundbreaking work earned him the Nobel Prize in Physics in 1939.
How does a Cyclotron Work?
The process begins when charged particles like positive or negative ions are injected into the centre of the cyclotron, where they start to move outward in a spiral path.
Inside the cyclotron, are two hollow, D-shaped metal electrodes called ‘dees’, placed between the poles of a large magnet. The magnetic field forces the particles into a circular path, while an alternating electric field boosts the particle’s energy every time it crosses the gap between two dees. As the particles gain speed and energy, they continue to spiral outward.
Once the particles reach the outer edge of the cyclotron, they are directed toward a target. When the accelerated particles collide with the target, they can cause nuclear reactions, producing radioactive isotopes.
Nearly a century after their invention, cyclotrons remain in high demand because of their reliability, efficiency, and versatility.
What’s the Difference Between Cyclotrons and other Particle Accelerators?
Particle accelerators have many applications in medicine, industry and research. These machines accelerate charged particles, such as electrons and protons, to high speeds, sometimes even close to the speed of light.
While all particle accelerators share a common goal - boosting the energy of particles - they achieve this in different ways.
Cyclotrons accelerate particles in a spiral path using a constant magnetic field and an alternating electric field. The spiral design is one of the cyclotron’s main advantages. It allows for continuous acceleration in a relatively small space. As a result, cyclotrons are typically smaller, often room-sized, and more affordable than other accelerators. They can be installed in hospitals or university labs without needing massive facilities. Cyclotrons are also well-suited for producing specific types of radioactive isotopes needed in medical imaging and cancer treatment, and for other localized applications in research or industry.
In contrast, linear accelerators, or linacs, propel particles in a straight line using a series of electric fields. While linacs can be simpler in design, they often require much more space to achieve the same energy levels as a cyclotron. They are commonly used in radiotherapy, where precise targeted beams of radiation are used to treat tumours.
Another type of accelerator is the synchrotron - a much larger and more complex machine found in national research centres. Like cyclotrons, they guide particles in a circular path, but with variable magnetic fields and radiofrequency acceleration. These machines can reach extremely high energies, making them suitable for research in particle physics, materials science, and even drug development. However, due to their size and cost, they are typically used by national or international research centres, not hospitals or small labs.
Details
A cyclotron is a type of particle accelerator invented by Ernest Lawrence in 1929–1930 at the University of California, Berkeley, and patented in 1932. A cyclotron accelerates charged particles outwards from the center of a flat cylindrical vacuum chamber along a spiral path. The particles are held to a spiral trajectory by a static magnetic field and accelerated by a rapidly varying electric field. Lawrence was awarded the 1939 Nobel Prize in Physics for this invention.
The cyclotron was the first "cyclical" accelerator. The primary accelerators before the development of the cyclotron were electrostatic accelerators, such as the math–Walton generator and the Van de Graaff generator. In these accelerators, particles would cross an accelerating electric field only once. Thus, the energy gained by the particles was limited by the maximum electrical potential that could be achieved across the accelerating region. This potential was in turn limited by electrostatic breakdown to a few million volts. In a cyclotron, by contrast, the particles encounter the accelerating region many times by following a spiral path, so the output energy can be many times the energy gained in a single accelerating step.
Cyclotrons were the most powerful particle accelerator technology until the 1950s, when they were surpassed by the synchrotron. Nonetheless, they are still widely used to produce particle beams for nuclear medicine and basic research. As of 2020, close to 1,500 cyclotrons were in use worldwide for the production of radionuclides for nuclear medicine and ultimately, for the production of radiopharmaceuticals. In addition, cyclotrons can be used for particle therapy, where particle beams are directly applied to patients.
Usage:
Basic research
For several decades, cyclotrons were the best source of high-energy beams for nuclear physics experiments. With the advent of strong focusing synchrotrons, cyclotrons were supplanted as the accelerators capable of producing the highest energies. However, due to their compactness, and therefore lower expense compared to high-energy synchrotrons, cyclotrons are still used to create beams for research where the primary consideration is not achieving the maximum possible energy. Cyclotron-based nuclear physics experiments are used to measure basic properties of isotopes (particularly short lived radioactive isotopes) including half-life, mass, interaction cross sections, and decay schemes.
Medical uses:
Radioisotope production
Cyclotron beams can be used to bombard other atoms to produce short-lived isotopes with a variety of medical uses, including medical imaging and radiotherapy. Positron and gamma emitting isotopes, such as fluorine-18, carbon-11, and technetium-99m are used for PET and SPECT imaging. While cyclotron produced radioisotopes are widely used for diagnostic purposes, therapeutic uses are still largely in development. Proposed isotopes include astatine-211, palladium-103, rhenium-186, and bromine-77, among others.
Beam therapy
The first suggestion that energetic protons could be an effective treatment method was made by Robert R. Wilson in a paper published in 1946 while he was involved in the design of the Harvard Cyclotron Laboratory.
Beams from cyclotrons can be used in particle therapy to treat cancer. Ion beams from cyclotrons can be used, as in proton therapy, to penetrate the body and kill tumors by radiation damage, while minimizing damage to healthy tissue along their path.
As of 2020, there were approximately 80 facilities worldwide for radiotherapy using beams of protons and heavy ions, consisting of a mixture of cyclotrons and synchrotrons. Cyclotrons are primarily used for proton beams, while synchrotrons are used to produce heavier ions.
PET : Positron Emission Tomography.
SPECT : Single Photon Emission Computed Tomography.
Advantages and limitations
Livingston and Lawrence with the 69 cm (27 in) cyclotron at the Lawrence Radiation Laboratory. The metal arch supports the magnet's core, and the large cylindrical boxes contain the coils of wire that generate the magnetic field. The vacuum chamber containing the "dee" electrodes is in the center between the magnet's poles.
The most obvious advantage of a cyclotron over a linear accelerator is that because the same accelerating gap is used many times, it is both more space efficient and more cost efficient; particles can be brought to higher energies in less space, and with less equipment. The compactness of the cyclotron reduces other costs as well, such as foundations, radiation shielding, and the enclosing building. Cyclotrons have a single electrical driver, which saves both equipment and power costs. Furthermore, cyclotrons are able to produce a continuous beam of particles at the target, so the average power passed from a particle beam into a target is relatively high compared to the pulsed beam of a synchrotron.
However, as discussed above, a constant frequency acceleration method is only possible when the accelerated particles are approximately obeying Newton's laws of motion. If the particles become fast enough that relativistic effects become important, the beam slips out of phase with the oscillating electric field, and cannot receive any additional acceleration. The classical cyclotron (constant field and frequency) is therefore only capable of accelerating particles up to a few percent of the speed of light. Synchro-, isochronous, and other types of cyclotrons can overcome this limitation, with the tradeoff of increased complexity and cost.
An additional limitation of cyclotrons is due to space charge effects – the mutual repulsion of the particles in the beam. As the amount of particles (beam current) in a cyclotron beam is increased, the effects of electrostatic repulsion grow stronger until they disrupt the orbits of neighboring particles. This puts a functional limit on the beam intensity, or the number of particles which can be accelerated at one time, as distinct from their energy.
Additional Information
The cyclotron was one of the earliest types of particle accelerators, and is still used as the first stage of some large multi-stage particle accelerators. It makes use of the magnetic force on a moving charge to bend moving charges into a semicircular path between accelerations by an applied electric field. The applied electric field accelerates electrons between the "dees" of the magnetic field region. The field is reversed at the cyclotron frequency to accelerate the electrons back across the gap.
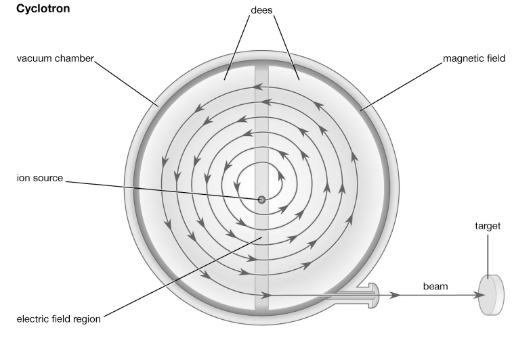
#298 Science HQ » Intramuscular Injection » 2026-02-24 22:51:05
- Jai Ganesh
- Replies: 0
Intramuscular Injection
Gist
Intramuscular (IM) injections deliver medication deep into muscle tissue for rapid systemic absorption, usually in volumes of 2–5 mL. Common sites include the deltoid (arm), vastus lateralis (thigh), and ventrogluteal (hip) muscles. Injections require a 90-degree angle, proper site identification, and sterile technique to avoid complications like pain, infection, or nerve damage.
Intramuscular (IM) injections deliver medicine deep into muscle tissue for fast absorption, used for vaccines (like flu shots, MMR), antibiotics, hormones (like Depo-Provera), emergency meds (epinephrine, glucagon), and antipsychotics, especially when oral intake is impossible or less effective, providing quicker action than oral routes or sustained release. Common sites include the thigh, upper arm (deltoid), and buttocks, chosen for adequate muscle and to avoid nerves/vessels, allowing for rapid systemic effect or controlled long-term release.
Summary
Intramuscular injection (IM) is installing medications into the depth of specifically selected muscles. The bulky muscles have good vascularity, and therefore the injected drug quickly reaches the systemic circulation and thereafter into the specific region of action, bypassing the first-pass metabolism. It is one of the most common medical procedures to be performed annually.
Intramuscular injection (IM) is installing medications into the depth of specifically selected muscles. The bulky muscles have good vascularity, and therefore the injected drug quickly reaches the systemic circulation and thereafter into the specific region of action, bypassing the first-pass metabolism. It is one of the most common medical procedures to be performed annually. However, there is still a lack of adherence to recommended guidelines and an algorithm for giving IM among health professionals worldwide.
Drugs may be given intramuscularly both for prophylactic (around 5% for immunization) as well as curative purposes (accounting for more than 95% of IM injections).
The most common medications given by IM route include:
* Antibiotics- penicillin G benzathine penicillin, streptomycin
* Biologicals- immunoglobins, vaccines, and toxoids
* Hormonal agents- testosterone, medroxyprogesterone
Any nonirritant and soluble drugs may be given IM during an emergency scenario.
Details
Intramuscular injection, often abbreviated IM, is the injection of a substance into a muscle. In medicine, it is one of several methods for parenteral administration of medications. Intramuscular injection may be preferred because muscles have larger and more numerous blood vessels than subcutaneous tissue, leading to faster absorption than subcutaneous or intradermal injections. Medication administered via intramuscular injection is not subject to the first-pass metabolism effect which affects oral medications.
Common sites for intramuscular injections include the deltoid muscle of the upper arm and the gluteal muscle of the buttock. In infants, the vastus lateralis muscle of the thigh is commonly used. The injection site must be cleaned before administering the injection, and the injection is then administered in a fast, darting motion to decrease the discomfort to the individual. The volume to be injected in the muscle is usually limited to 2–5 milliliters, depending on injection site. A site with signs of infection or muscle atrophy should not be chosen. Intramuscular injections should not be used in people with myopathies or those with trouble clotting.
Intramuscular injections commonly result in pain, redness, and swelling or inflammation around the injection site. These side effects are generally mild and last no more than a few days at most. Rarely, nerves or blood vessels around the injection site can be damaged, resulting in severe pain or paralysis. If proper technique is not followed, intramuscular injections can result in localized infections such as abscesses and gangrene. While historically aspiration, or pulling back on the syringe before injection, was recommended to prevent inadvertent administration into a vein, it is no longer recommended for most injection sites by some countries.
Uses
Intramuscular injection is commonly used for medication administration. Medication administered in the muscle is generally quickly absorbed in the bloodstream, and avoids the first pass metabolism which occurs with oral administration. The medication may not be considered 100% bioavailable as it must still be absorbed from the muscle, which occurs over time. An intramuscular injection is less invasive than an intravenous injection and also generally takes less time, as the site of injection (a muscle versus a vein) is much larger. Medications administered in the muscle may also be administered as depot injections, which provide slow, continuous release of medicine over a longer period of time. Certain substances, including ketamine, may be injected intramuscularly for recreational purposes. Disadvantages of intramuscular administration include skill and technique required, pain from injection, anxiety or fear (especially in children), and difficulty in self-administration which limits its use in outpatient medicine.
Vaccines, especially inactivated vaccines, are commonly administered via intramuscular injection. However, it has been estimated that for every vaccine injected intramuscularly, 20 injections are given to administer drugs or other therapy. This can include medications such as antibiotics, immunoglobulin, and hormones such as testosterone and medroxyprogesterone. In a case of severe allergic reaction, or anaphylaxis, a person may use an epinephrine autoinjector to self-administer epinephrine into the muscle.
Contraindications
Because an intramuscular injection can be used to administer many types of medications, specific contraindications depend in large part on the medication being administered. Injections of medications are necessarily more invasive than other forms of administration such as by mouth or topical and require training to perform appropriately, without which complications can arise regardless of the medication being administered. For this reason, unless there are desired differences in rate of absorption, time to onset, or other pharmacokinetic parameters in the specific situation, a less invasive form of drug administration (usually by mouth) is preferred.
Intramuscular injections are generally avoided in people with low platelet count or clotting problems, to prevent harm due to potential damage to blood vessels during the injection. They are also not recommended in people who are in hypovolemic shock, or have myopathy or muscle atrophy, as these conditions may alter the absorption of the medication. The damage to the muscle caused by an intramuscular injections may interfere with the accuracy of certain cardiac tests for people with suspected myocardial infarction and for this reason other methods of administration are preferred in such instances. In people with an active myocardial infarction, the decrease in circulation may result in slower absorption from an IM injection. Specific sites of administration may also be contraindicated if the desired injection site has an infection, swelling, or inflammation. Within a specific site of administration, the injection should not be given directly over irritation or redness, birthmarks or moles, or areas with scar tissue.
Risks and complications
As an injection necessitates piercing the skin, there is a risk of infection from bacteria or other organisms present in the environment or on the skin before the injection. This risk is minimized by using proper aseptic technique in preparing the injection and sanitizing the injection site before administration. Intramuscular injections may also cause an abscess or gangrene at the injection site, depending on the specific medication and amount administered. There is also a risk of nerve or vascular injury if a nerve or blood vessel is inadvertently hit during injection. If single-use or sterilized equipment is not used, there is the risk of transmission of infectious disease between users, or to a practitioner who inadvertently injures themselves with a used needle, termed a needlestick injury.
Site-specific complications
Injections into the deltoid site in the arm can result in unintentional damage to the radial and axillary nerves. In rare cases when not performed properly, the injection may result in shoulder dysfunction. The most frequent complications of a deltoid injection include pain, redness, and inflammation around the injection site, which are almost always mild and last only a few days at most.
The dorsogluteal site of injection is associated with a higher risk of skin and tissue trauma, muscle fibrosis or contracture, hematoma, nerve palsy, paralysis, and infections such as abscesses and gangrene. Furthermore, injection in the gluteal muscle poses a risk for damage to the sciatic nerve, which may cause shooting pain or a sensation of burning. Sciatic nerve damage can also affect a person's ability to move their foot on the affected side, and other parts of the body controlled by the nerve. Damage to the sciatic nerve can be prevented by using the ventrogluteal site instead, and by selecting an appropriate size and length of needle for the injection.
Additional Information
An intramuscular injection is a technique for delivering medication deep into the muscles. This allows the medication to absorb quickly into the bloodstream.
You may have received an intramuscular injection at a doctor’s office the last time you got a vaccine, like the flu shot.
In some cases, a person may also self-administer an intramuscular injection. For example, certain drugs that treat multiple sclerosis or rheumatoid arthritis may require self-injection.
What are intramuscular injections used for?
Intramuscular injections are a common practice in modern medicine. They’re used to deliver drugs and vaccines. Several drugs and almost all injectable vaccines are delivered this way.
Intramuscular injections are used when other types of delivery methods are not recommended. These include:
oral (swallowed into the stomach)
intravenous (injected into the vein)
subcutaneous (injected into the fatty tissue just under the layer of skin)
Intramuscular injections may sometimes be used instead of intravenous injections because some drugs are irritating to veins or because a suitable vein cannot be located. However, not all intravenous medications can be administered intramuscularly.
They may be used instead of oral delivery because some drugs are destroyed by the digestive system when you swallow them.
Intramuscular injections are absorbed faster than subcutaneous injections. This is because muscle tissue has a greater blood supply than the tissue just under your skin. Muscle tissue can also hold a larger volume of medication than subcutaneous tissue.
Intramuscular injection sites
Intramuscular injections are often given in the following areas:
* Deltoid muscle of the arm
The deltoid muscle is the site most typically used for vaccines. However, this site is not common for self-injection because its small muscle mass limits the volume of medication that can be injected — typically no more than 1 milliliter.
It’s also challenging to use this site for self-injection. A caregiver, friend, or family member can assist with injections into this muscle.
To locate this site, feel for the bone (acromion process) located at the top of your upper arm. The correct area to give the injection is two finger widths below the acromion process. At the bottom of the two fingers will be an upside-down triangle. Give the injection in the center of the triangle.
Vastus lateralis muscle of the thigh
The thigh may be used when the other sites are not available or if you need to administer the medication on your own.
Divide the upper thigh into three equal parts. Locate the middle of these three sections.
Ventrogluteal muscle of the hip
The ventrogluteal muscle is the safest site for adults and children older than 7 months. It’s deep and not close to any major blood vessels or nerves. However, this site is difficult for self-injection and may require the help of a friend, family member, or caregiver.
Place the heel of your hand on the hip of the person receiving the injection, with your fingers pointing toward their head. Position your fingers so your thumb points toward their groin, and you feel the pelvis under your pinky finger. Spread your index and middle fingers in a slight V shape, and inject the needle into the middle of that V.
#299 Re: Jai Ganesh's Puzzles » General Quiz » 2026-02-24 22:14:31
Hi,
#10765. What does the term in Biology Genus mean?
#10766. What does the term in Biology Gizzard mean?
#300 Re: Jai Ganesh's Puzzles » English language puzzles » 2026-02-24 21:59:08
Hi,
#5961. What does the noun funfair mean?
#5962. What does the adjective fungible mean?