Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
- Index
- » This is Cool
- » Magma
Pages: 1
#1 2025-07-21 17:42:25
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 52,349
Magma
Magma
Gist
Magma is melted rock or rock in liquid form. However, magma is the specific term used to denote liquid rock that is within Earth's interior, or below Earth's surface.
Some examples of magma are felsic and mafic magma. Both felsic and mafic magmas are high in silicon content. However, felsic magma is higher in silica content and is more viscous or sticky. It typically solidifies into igneous rock like rhyolite.
Mafic magma has a slightly lower silica content and is much less viscous than felsic. Mafic magma typically solidifies into igneous rock like basalt.
Magma is defined as a composite fluid material that consists of solid minerals, gas bubbles, and a silicate melt matrix, formed by partial melting within the Earth. Its composition varies and is primarily derived from the mantle or crust, resulting in different types such as basalts and granites.
Magma is extremely hot liquid and semi-liquid rock located under Earth's surface. Earth has a layered structure that consists of the inner core, outer core, mantle, and crust. Much of the planet's mantle consists of magma. This magma can push through holes or cracks in the crust, causing a volcanic eruption.
Summary
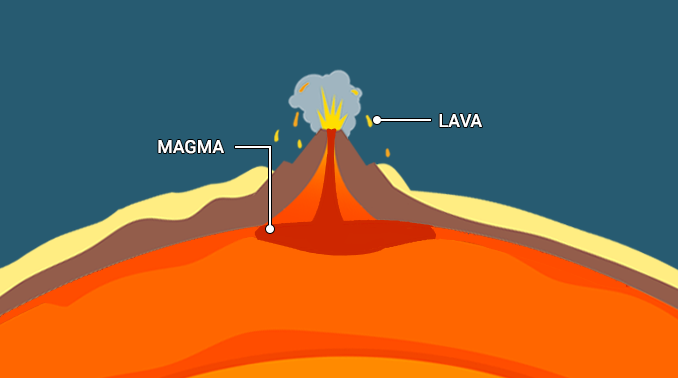
Magma is extremely hot liquid and semi-liquid rock located under Earth’s surface. When magma flows onto Earth’s surface, it is called lava.
Magma is extremely hot liquid and semi-liquid rock located under Earth’s surface. Earth has a layered structure that consists of the inner core, outer core, mantle, and crust. Much of the planet’s mantle consists of magma. This magma can push through holes or cracks in the crust, causing a volcanic eruption. When magma flows or erupts onto Earth’s surface, it is called lava.
Like solid rock, magma is a mixture of minerals. It also contains small amounts of dissolved gases such as water vapor, carbon dioxide, and sulfur. The high temperatures and pressure under Earth’s crust keep magma in its fluid state.
There are three basic types of magma: basaltic, andesitic, and rhyolitic, each of which has a different mineral composition. All types of magma have a significant percentage of silicon dioxide. Basaltic magma is high in iron, magnesium, and calcium but low in potassium and sodium. It ranges in temperature from about 1000oC to 1200oC (1832oF to 2192oF). Andesitic magma has moderate amounts of these minerals, with a temperature range from about 800oC to 1000oC (1472oF to 1832oF). Rhyolitic magma is high in potassium and sodium but low in iron, magnesium, and calcium. It occurs in the temperature range of about 650oC to 800oC (1202oF to 1472oF). Both the temperature and mineral content of magma affect how easily it flows.
The viscosity (thickness) of the magma that erupts from a volcano affects the shape of the volcano. Volcanoes with steep slopes tend to form from very viscous magma, while flatter volcanoes form from magma that flows easily.
Details
Magma is the molten or semi-molten natural material from which all igneous rocks are formed. Magma (sometimes colloquially but incorrectly referred to as lava) is found beneath the surface of the Earth, and evidence of magmatism has also been discovered on other terrestrial planets and some natural satellites. Besides molten rock, magma may also contain suspended crystals and gas bubbles.
Magma is produced by melting of the mantle or the crust in various tectonic settings, which on Earth include subduction zones, continental rift zones, mid-ocean ridges and hotspots. Mantle and crustal melts migrate upwards through the crust where they are thought to be stored in magma chambers or trans-crustal crystal-rich mush zones. During magma's storage in the crust, its composition may be modified by fractional crystallization, contamination with crustal melts, magma mixing, and degassing. Following its ascent through the crust, magma may feed a volcano and be extruded as lava, or it may solidify underground to form an intrusion, such as a dike, a sill, a laccolith, a pluton, or a batholith.
While the study of magma has relied on observing magma after its transition into a lava flow, magma has been encountered in situ three times during geothermal drilling projects, twice in Iceland (see Use in energy production) and once in Hawaii.
Physical and chemical properties
Magma consists of liquid rock that usually contains suspended solid crystals. As magma approaches the surface and the overburden pressure drops, dissolved gases bubble out of the liquid, so that magma near the surface consists of materials in solid, liquid, and gas phases.
Composition
Most magma is rich in silica. Rare nonsilicate magma can form by local melting of nonsilicate mineral deposits or by separation of a magma into separate immiscible silicate and nonsilicate liquid phases.
Silicate magmas are molten mixtures dominated by oxygen and silicon, the most abundant chemical elements in the Earth's crust, with smaller quantities of aluminium, calcium, magnesium, iron, sodium, and potassium, and minor amounts of many other elements. Petrologists routinely express the composition of a silicate magma in terms of the weight or molar mass fraction of the oxides of the major elements (other than oxygen) present in the magma.
Because many of the properties of a magma (such as its viscosity and temperature) are observed to correlate with silica content, silicate magmas are divided into four chemical types based on silica content: felsic, intermediate, mafic, and ultramafic.
Evolution of magmas
Most magmas are fully melted only for small parts of their histories. More typically, they are mixes of melt and crystals, and sometimes also of gas bubbles. Melt, crystals, and bubbles usually have different densities, and so they can separate as magmas evolve.
As magma cools, minerals typically crystallize from the melt at different temperatures. This resembles the original melting process in reverse. However, because the melt has usually separated from its original source rock and moved to a shallower depth, the reverse process of crystallization is not precisely identical. For example, if a melt was 50% each of diopside and anorthite, then anorthite would begin crystallizing from the melt at a temperature somewhat higher than the eutectic temperature of 1274 °C. This shifts the remaining melt towards its eutectic composition of 43% diopside. The eutectic is reached at 1274 °C, the temperature at which diopside and anorthite begin crystallizing together. If the melt was 90% diopside, the diopside would begin crystallizing first until the eutectic was reached.
If the crystals remained suspended in the melt, the crystallization process would not change the overall composition of the melt plus solid minerals. This situation is described as equillibrium crystallization. However, in a series of experiments culminating in his 1915 paper, Crystallization-differentiation in silicate liquids, Norman L. Bowen demonstrated that crystals of olivine and diopside that crystallized out of a cooling melt of forsterite, diopside, and silica would sink through the melt on geologically relevant time scales. Geologists subsequently found considerable field evidence of such fractional crystallization.
When crystals separate from a magma, then the residual magma will differ in composition from the parent magma. For instance, a magma of gabbroic composition can produce a residual melt of granitic composition if early formed crystals are separated from the magma. Gabbro may have a liquidus temperature near 1,200 °C, and the derivative granite-composition melt may have a liquidus temperature as low as about 700 °C. Incompatible elements are concentrated in the last residues of magma during fractional crystallization and in the first melts produced during partial melting: either process can form the magma that crystallizes to pegmatite, a rock type commonly enriched in incompatible elements. Bowen's reaction series is important for understanding the idealised sequence of fractional crystallisation of a magma.
Magma composition can be determined by processes other than partial melting and fractional crystallization. For instance, magmas commonly interact with rocks they intrude, both by melting those rocks and by reacting with them. Assimilation near the roof of a magma chamber and fractional crystallization near its base can even take place simultaneously. Magmas of different compositions can mix with one another. In rare cases, melts can separate into two immiscible melts of contrasting compositions.
Additional Information
Magma, molten or partially molten rock from which igneous rocks form. It usually consists of silicate liquid, although carbonate and sulfide melts occur as well. Magma migrates either at depth or to Earth’s surface and is ejected as lava. Suspended crystals and fragments of unmelted rock may be transported in the magma; dissolved volatiles may separate as bubbles and some liquid may crystallize during movement. Several interrelated physical properties determine the characteristics of magma, including chemical composition, viscosity, dissolved gases, and temperature.
Magma reaches Earth's surface as lava and flows over Hawaiian vegetation and into the oceanMolten, or hot liquefied, rock located deep below Earth's surface is called magma. When a volcano erupts, the magma rises and either gently flows onto the surface or is violently shot into the air. When magma reaches Earth's surface, it usually mixes with gases, and it is called lava. Fresh lava ranges from 1,300 to 2,200 °F (700 to 1,200 °C) and glows red hot to white hot as it flows. As lava cools, it solidifies to form rock.
There are three major types of magma. Basaltic (or mafic) magma predominates in nonexplosive volcanic eruptions. It is a high-temperature magma (1,200 °C [about 2,200 °F]) characterized by flowing lava, and it is made up of about 45–55 percent silica (SiO2) by weight. In contrast, the temperature of rhyolitic (or felsic) magma is much lower (750–850 °C [about 1,400–1,560 °F]), but its silica content is higher, ranging from about 65 to 75 percent by weight. Rhyolitic magmas are characteristic of the most explosive eruptions, which also produce ash falls and pyroclastic flows. Andesitic magma is intermediate in temperature (800–1,000 °C [about 1,470–1,830 °F]) and silica content. (See also felsic and mafic rocks.)
As magma cools, crystals form in a systematic manner, which is most simply expressed in the form of Bowen’s reaction series; early high-temperature crystals will tend to react with the liquid to form other minerals at lower temperatures. Two series are recognized: (1) a discontinuous reaction series, which from high to low temperatures is composed of olivine, orthopyroxene, clinopyroxene, amphibole, and biotite; and (2) a continuous reaction series, represented by high-temperature calcium-rich plagioclase to low-temperature sodium-rich plagioclase. Numerous variations can occur during crystallization to influence the resulting rock. Such variations include separation of early crystals from liquid, preventing a reaction; cooling of magma too rapidly for reactions to occur; and loss of volatiles, which may remove some components from the magma. Transport and emplacement of magma is strongly affected by its viscosity and by the fracture characteristics of rocks through which it moves. Viscosity is reduced by water and a lower silica content.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1
- Index
- » This is Cool
- » Magma