Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
- Index
- » Science HQ
- » Erbium
Pages: 1
#1 2025-08-25 16:46:33
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 51,538
Erbium
Erbium
Gist
Erbium (Er) is a rare-earth, f-block element with atomic number 68, found as a silvery-white solid metal when isolated. It is used to color glass, in metal alloys, and as burnable poison in nuclear reactors. Due to its optical properties, erbium is also crucial for lasers in medical treatments (like resurfacing and tattoo removal) and for amplifying signals in long-distance fiber optic communications.
This metal is used to colour the lenses of glasses. It is also used in the control rods of nuclear reactors. It has a vital application in the production of metal alloys. Erbium ion's unique wavelength aids laser surgery.
Summary
Erbium is a chemical element; it has symbol Er and atomic number 68. A silvery-white solid metal when artificially isolated, natural erbium is always found in chemical combination with other elements. It is a lanthanide, a rare-earth element, originally found in the gadolinite mine in Ytterby, Sweden, which is the source of the element's name.
Erbium's principal uses involve its pink-colored Er3+ ions, which have optical fluorescent properties particularly useful in certain laser applications. Erbium-doped glasses or crystals can be used as optical amplification media, where Er3+ ions are optically pumped at around 980 or 1480 nm and then radiate light at 1530 nm in stimulated emission. This process results in an unusually mechanically simple laser optical amplifier for signals transmitted by fiber optics. The 1550 nm wavelength is especially important for optical communications because standard single mode optical fibers have minimal loss at this particular wavelength.
In addition to optical fiber amplifier-lasers, a large variety of medical applications (e.g. dermatology, dentistry) rely on the erbium ion's 2940 nm emission (see Er:YAG laser) when lit at another wavelength, which is highly absorbed in water in tissues, making its effect very superficial. Such shallow tissue deposition of laser energy is helpful in laser surgery, and for the efficient production of steam which produces enamel ablation by common types of dental laser.
Details
Erbium (Er) is a chemical element, a rare-earth metal of the lanthanide series of the periodic table.
Pure erbium is a silvery white metal that is relatively stable in air. It slowly reacts with water and quickly dissolves in diluted acids, except hydrofluoric acid (HF) because of formation of the protective fluoride (ErF3) layer on the surface of the metal. Erbium is a very strong paramagnet above approximately 85 K (−188 °C, or −307 °F). Between 85 K and 20 K (−253 °C, or −424 °F) the metal is antiferromagnetic, and below about 20 K it is arranged in a conical ferromagnetic structure.
The element was discovered in 1842 as an oxide by Carl Gustaf Mosander, who originally called it terbia; in the confusion arising from the similarity in the properties of the rare-earth elements, the names of two, terbium and erbium, became interchanged (c. 1860). The element occurs in many rare-earth minerals; among the more important are the laterite ionic clays, xenotime, and euxenite. Erbium also occurs in the products of nuclear fission. In Earth’s crust, erbium is as abundant as tantalum and tungsten.
Natural erbium is a mixture of six stable isotopes: erbium-166 (33.5 percent), erbium-168 (26.98 percent), erbium-167 (22.87 percent), erbium-170 (14.91 percent), erbium-164 (1.6 percent), and erbium-162 (0.14 percent). Not counting nuclear isomers, a total of 30 radioactive isotopes of erbium are known. Their mass varies from 142 to 177. All the radioactive isotopes of erbium are relatively unstable: their half-lives range from 1 second (erbium-145) to 9.4 days (erbium-169).
Commercial purification is accomplished by liquid-liquid solvent extraction and ion-exchange methods. The metal itself is prepared by metallothermic reduction of the anhydrous fluoride with calcium. Only one allotropic (structural) form is known for erbium. The element adopts a close-packed hexagonal structure with a = 3.5592 Å and c = 5.5850 Å at room temperature.
When raised to a high-energy state by absorption of infrared light, the Er3+ ion emits photons at wavelengths of 1.55 micrometres—one of the wavelengths commonly employed in fibre-optic signal transmission. Hence, the major use of erbium is in fibre-optic telecommunications as a component of the signal amplifiers in long-distance telephone and data cables. Its compounds are used in lasers and as a pink colouring agent for glasses. Erbium-stabilized zirconia (ZrO2) makes pink synthetic gems. Another small-scale use of erbium is in the intermetallic compound Er3Ni, which has a high magnetic heat capacity around 4 K (−269 °C, or −452 °F), which is needed for effective regenerative heat exchange at low temperatures, and, therefore, the compound is employed as a regenerator material in low-temperature cryocoolers.
Erbium behaves as a typical rare-earth element, forming compounds in which its oxidation state is +3, such as the pink oxide Er2O3. The Er3+ ion is pink in solution.
Element Properties
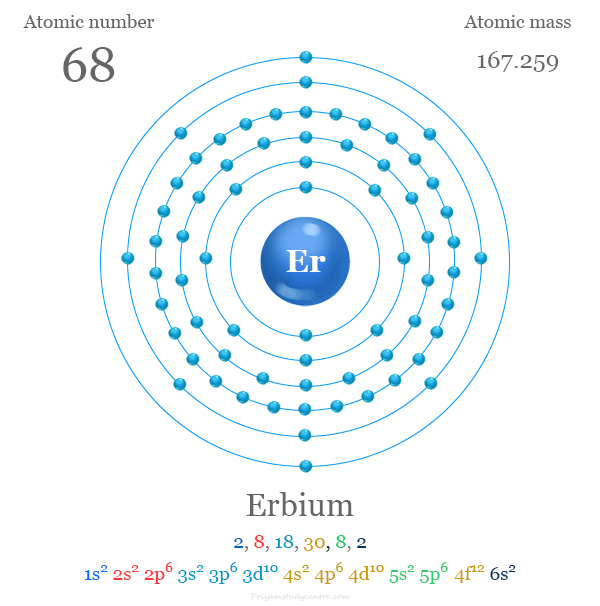
atomic number : 68
atomic weight : 167.259
melting point : 1,529 °C (2,784 °F)
boiling point : 2,868 °C (5,194 °F)
specific gravity : 9.066 (24 °C, or 75 °F)
oxidation state : +3.
Additional Information:
Appearance
A soft, silvery metallic element.
Uses
Erbium finds little use as a metal because it slowly tarnishes in air and is attacked by water.
When alloyed with metals such as vanadium, erbium lowers their hardness and improves their workability.
Erbium oxide is occasionally used in infrared absorbing glass, for example safety glasses for welders and metal workers. When erbium is added to glass it gives the glass a pink tinge. It is used to give colour to some sunglasses and imitation gems.
Broadband signals, carried by fibre optic cables, are amplified by including erbium in the glass fibre.
Biological role
Erbium has no known biological role, and has low toxicity.
Natural abundance
Erbium is found principally in the minerals monazite and bastnaesite. It can be extracted by ion exchange and solvent extraction.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1
- Index
- » Science HQ
- » Erbium