Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
- Index
- » Science HQ
- » Ruthenium
Pages: 1
#1 2025-08-06 16:23:14
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 51,538
Ruthenium
Ruthenium
Gist
Ruthenium is defined as a critical rare transition metal belonging to the platinum group, widely utilized in material science, catalysis, medicine, and energy applications such as solar cells and batteries.
Ruthenium is primarily used as an alloying agent. Adding 0.1% ruthenium to titanium makes titanium 100 times more resistant to corrosion. Small amounts of ruthenium are added to platinum and palladium to strengthen them.
Summary
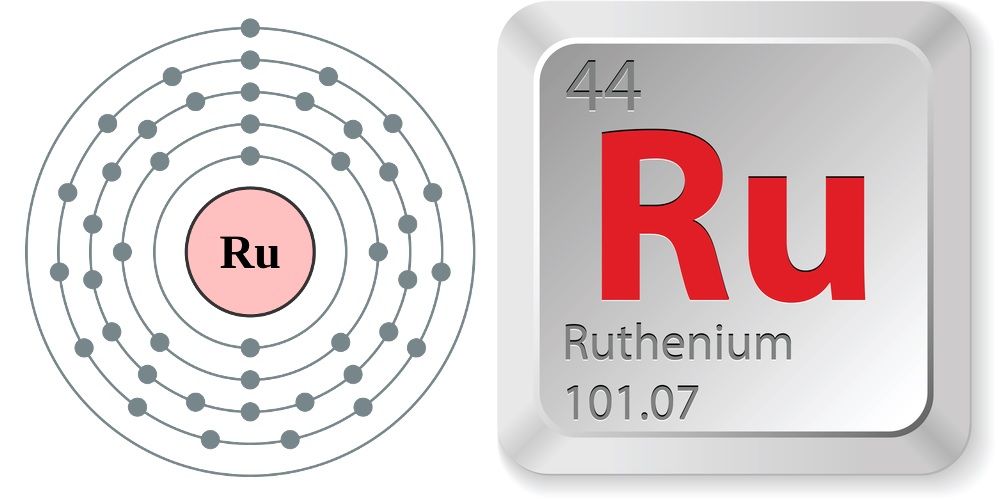
Ruthenium is a chemical element; it has symbol Ru and atomic number 44. It is a rare transition metal belonging to the platinum group of the periodic table. Like the other metals of the platinum group, ruthenium is unreactive to most chemicals. Karl Ernst Claus, a Russian scientist of Baltic-German ancestry, discovered the element in 1844 at Kazan State University and named it in honor of Russia, using the Latin name Ruthenia. Ruthenium is usually found as a minor component of platinum ores; the annual production has risen from about 19 tonnes in 2009 to some 35.5 tonnes in 2017. Most ruthenium produced is used in wear-resistant electrical contacts and thick-film resistors. A minor application for ruthenium is in platinum alloys and as a chemical catalyst. A new application of ruthenium is as the capping layer for extreme ultraviolet photomasks. Ruthenium is generally found in ores with the other platinum group metals in the Ural Mountains and in North and South America. Small but commercially important quantities are also found in pentlandite extracted from Sudbury, Ontario, and in pyroxenite deposits in South Africa.
Details
Ruthenium (Ru) is a chemical element, one of the platinum metals of Groups 8–10 (VIIIb), Periods 5 and 6, of the periodic table, used as an alloying agent to harden platinum and palladium. Silver-gray ruthenium metal looks like platinum but is rarer, harder, and more brittle. The Russian chemist Karl Karlovich Klaus established (1844) the existence of this rare, bright metal and retained the name his countryman Gottfried Wilhelm Osann had suggested (1828) for a platinum-group element whose discovery had remained inconclusive. Ruthenium has a low crustal abundance of about 0.001 part per million. Elemental ruthenium occurs in native alloys of iridium and osmium, along with the other platinum metals: up to 14.1 percent in iridosmine and 18.3 percent in siserskite. It also occurs in sulfide and other ores (e.g., in pentlandite of the Sudbury, Ontario, Canada, nickel-mining region) in very small quantities that are commercially recovered.
Because of its high melting point, ruthenium is not easily cast; its brittleness, even at white heat, makes it very difficult to roll or draw into wires. Thus, the industrial application of metallic ruthenium is restricted to use as an alloy for platinum and other metals of the platinum group. Processes for isolating it are an integral part of the metallurgical art that applies to all platinum metals. It serves the same function as iridium for the hardening of platinum and, in conjunction with rhodium, is used to harden palladium. Ruthenium-hardened alloys of platinum and palladium are superior to the pure metals in the manufacture of fine jewelry and of electrical contacts for wear resistance.
Ruthenium is found among the fission products of uranium and plutonium in nuclear reactors. Radioactive ruthenium-106 (one-year half-life) and its short-lived daughter rhodium-106 contribute an important fraction of the residual radiation in reactor fuels a year following their use. Recovery of the unused fissionable material is made difficult because of the radiation hazard and the chemical similarity between ruthenium and plutonium.
Natural ruthenium consists of a mixture of seven stable isotopes: ruthenium-96 (5.54 percent), ruthenium-98 (1.86 percent), ruthenium-99 (12.7 percent), ruthenium-100 (12.6 percent), ruthenium-101 (17.1 percent), ruthenium-102 (31.6 percent), and ruthenium-104 (18.6 percent). It has four allotropic forms. Ruthenium has a high resistance to chemical attack. Ruthenium is, with osmium, the most noble of the platinum metals; the metal does not tarnish in air at ordinary temperatures and resists attack by strong acids, even by aqua regia. Ruthenium is brought into soluble form by fusion with an alkaline oxidizing flux, such as sodium peroxide (Na2O2), especially if an oxidizing agent such as sodium chlorate is present.
The −2 and 0 through +8 states are known, but +2, +3, +4, +6, and +8 are most important. In addition to carbonyl and organometallic compounds in the low oxidation states −2, 0, and +1, ruthenium forms compounds in every oxidation state from +2 to +8. Very volatile ruthenium tetroxide, RuO4, used in separating ruthenium from other heavy metals, contains the element in the +8 oxidation state. (Although ruthenium tetroxide, RuO4, has similar stability and volatility to osmium tetroxide, OsO4, it differs in that it cannot be formed from the elements.) The chemistries of ruthenium and osmium are generally similar. The higher oxidation states +6 and +8 are much more readily obtained than for iron, and there is an extensive chemistry of the tetroxides, oxohalides, and oxo anions. There is little, if any, evidence that simple aquo ions exist, and virtually all its aqueous solutions, whatever the anions present, may be considered to contain complexes. Numerous coordination complexes are known, including a unique series of nitrosyl (NO) complexes.
Element Properties
atomic number : 44
atomic weight : 101.07
melting point : 2,250° C (4,082° F)
boiling point : 3,900° C (7,052° F)
specific gravity : 12.30 (20° C)
valence : 1, 2, 3, 4, 5, 6, 7, 8.
Additional Information:
Appearance
A shiny, silvery metal.
Uses
Many new uses are emerging for ruthenium. Most is used in the electronics industry for chip resistors and electrical contacts. Ruthenium oxide is used in the chemical industry to coat the anodes of electrochemical cells for chlorine production. Ruthenium is also used in catalysts for ammonia and acetic acid production. Ruthenium compounds can be used in solar cells, which turn light energy into electrical energy.
Ruthenium is one of the most effective hardeners for platinum and palladium, and is alloyed with these metals to make electrical contacts for severe wear resistance. It is used in some jewellery as an alloy with platinum.
Biological role
Ruthenium has no known biological role. Ruthenium(IV) oxide is highly toxic.
Natural abundance
Ruthenium is one of the rarest metals on Earth. It is found uncombined in nature; however, it is more commonly found associated with other platinum metals in the minerals pentlandite and pyroxinite. It is obtained commercially from the wastes of nickel refining.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1
- Index
- » Science HQ
- » Ruthenium