Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
- Index
- » Science HQ
- » Strontium
Pages: 1
#1 2025-07-31 17:56:57
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 51,538
Strontium
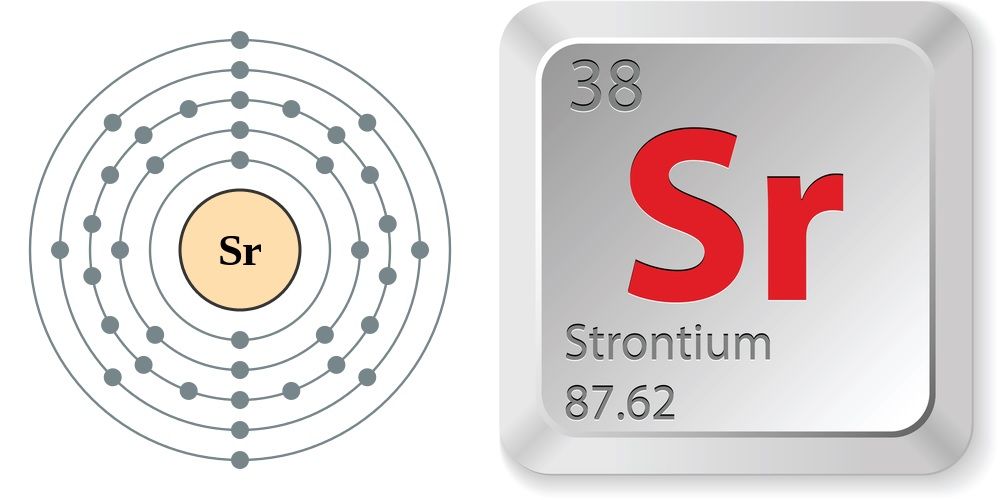
Strontium
Gist
Strontium (Sr) is a chemical element, specifically an alkaline earth metal, with atomic number 38 and atomic mass of approximately 87.62 g/mol. It's a soft, silvery-white metal that readily reacts with air and water, forming a dark oxide layer. Strontium shares many chemical and physical properties with calcium and barium, its neighboring elements on the periodic table.
Strontium (Sr) is a chemical element used in various applications, including fireworks, where it produces a vibrant red color, and in the manufacture of specialized glass for color televisions. It's also used in some dental products, like toothpaste for sensitive teeth, and in pharmaceuticals, particularly for treating osteoporosis. Additionally, strontium compounds are used in ceramics, paint pigments, fluorescent lights, and refining zinc.
Summary
Strontium is a chemical element; it has symbol Sr and atomic number 38. An alkaline earth metal, it is a soft silver-white yellowish metallic element that is highly chemically reactive. The metal forms a dark oxide layer when it is exposed to air. Strontium has physical and chemical properties similar to those of its two vertical neighbors in the periodic table, calcium and barium. It occurs naturally mainly in the minerals celestine and strontianite, and is mostly mined from these.
Both strontium and strontianite are named after Strontian, a village in Scotland near which the mineral was discovered in 1790 by Adair Crawford and William Cruickshank; it was identified as a new element the next year from its crimson-red flame test color. Strontium was first isolated as a metal in 1808 by Humphry Davy using the then newly discovered process of electrolysis. During the 19th century, strontium was mostly used in the production of sugar from sugar beets. At the peak of production of television cathode-ray tubes, as much as 75% of strontium consumption in the United States was used for the faceplate glass. With the replacement of cathode-ray tubes with other display methods, consumption of strontium has dramatically declined.
While natural strontium (which is mostly the isotope strontium-88) is stable, the synthetic strontium-90 is radioactive and is one of the most dangerous components of nuclear fallout, as strontium is absorbed by the body in a similar manner to calcium. Natural stable strontium, on the other hand, is not hazardous to health.
Details:
Strontium (Sr) is a chemical element, one of the alkaline-earth metals of Group 2 (IIa) of the periodic table. It is used as an ingredient in red signal flares and phosphors and is the principal health hazard in radioactive fallout.
Element Properties
atomic number : 38
atomic weight : 87.62
melting point : 769 °C (1,416 °F)
boiling point : 1,384 °C (2,523 °F)
specific gravity : 2.63
oxidation state : +2
Occurrence, properties, and uses
Strontium is a soft metal like lead and, when freshly cut, has a silvery lustre. It rapidly reacts in air to take on a yellowish colour; therefore, it must be protected from oxygen for storage. It does not occur free in nature. Although it is widely distributed with calcium, there are only two principal ores of strontium alone, celestine (SrSO4) and strontianite (SrCO3).
A mineral from a lead mine near the village of Strontian, in Argyll, Scotland, was originally misidentified as a type of barium carbonate, but Adair Crawford and William Cruickshank in 1789 noted that it was likely a different substance. The chemist Thomas Charles Hope named the new mineral strontites, after the village, and the corresponding “earth” (strontium oxide, SrO) was accordingly referred to as strontia. The metal was isolated (1808) by Sir Humphry Davy, who electrolyzed a mixture of the moist hydroxide or chloride with mercuric oxide, using a mercury cathode, and then evaporated the mercury from the resultant amalgam. He used the stem of the word strontia to form the name of the element.
Its cosmic abundance is estimated as 18.9 atoms (on a scale where the abundance of silicon = {10}^{6} atoms). It composes about 0.04 percent of Earth’s crust. The most important commercial source of strontium is celestine; more than two-thirds of the world’s supply comes from China, with Spain and Mexico supplying much of the remainder. Strontium may be obtained in the form of sticks by the contact cathode method of electrolysis, in which a cooled iron rod, acting as a cathode, just touches the surface of a fused mixture of potassium and strontium chlorides and is raised as the strontium solidifies on it. Metallic strontium may be also obtained by reduction of the oxide with aluminum. The metal is malleable and ductile and a good conductor of electricity, but there are relatively few uses for elemental strontium. One of them is as an alloying agent for aluminum or magnesium in cast engine blocks and wheels; the strontium improves the machinability and creep resistance of the metal.
Naturally occurring strontium is a mixture of four stable isotopes: strontium-88 (82.6 percent), strontium-86 (9.9 percent), strontium-87 (7.0 percent), and strontium-84 (0.56 percent). Depending on the location, it is possible for the ratios of strontium-87 to strontium-86 to differ by more than a factor of 5. This variation is used in dating geological samples and in identifying the provenance of skeletons and clay artifacts. About 16 synthetic radioactive isotopes have been produced by nuclear reactions, of which the longest-lived is strontium-90 (28.9-year half-life). This isotope, formed by nuclear explosions, is considered the most dangerous constituent of fallout. Because of its chemical resemblance to calcium, it is assimilated in bones and teeth, where it continues ejecting electrons that cause radiation injury by damaging bone marrow, impairing the process of forming new blood cells, and possibly inducing cancer. Under controlled conditions, however, it has been used for treatment of some superficial cancers and bone cancer. It is also used as a source in thickness gauges and has been used in radioisotope thermoelectric generators, where the heat of its radioactive decay is converted to electricity for long-lived, lightweight power sources in navigation buoys, remote weather stations, and space vehicles. Strontium-89 is employed in the treatment of bone cancer, as it targets bone tissues, delivers its beta radiation, and then decays in a few months’ time (half-life 51 days).
Strontium is not an essential element for higher life-forms, and its salts are generally nontoxic. The same “bone-seeking” property that makes strontium-90 dangerous is beneficially employed in strontium supplements to increase bone density and growth.
Compounds
In general, the chemistry of strontium is quite similar to that of calcium. In its compounds strontium has an exclusive oxidation state of +2, as the Sr2+ ion. The metal is an active reducing agent and readily reacts with halogens, oxygen, and sulfur to yield halides, oxide, and sulfide.
Strontium compounds have rather limited commercial value because the corresponding calcium and barium compounds generally serve the same purpose yet are cheaper. A few, however, have found application in industry and elsewhere. There is currently no substitute for the brilliant crimson colour produced by strontium salts such as strontium nitrate, Sr(NO3)2, and strontium chlorate, Sr(ClO3)2, in fireworks, flares, and tracer ammunition. About 5–10 percent of all strontium production is consumed in pyrotechnics. Strontium hydroxide, Sr(OH)2, is sometimes used to extract sugar from molasses because it forms a soluble saccharide from which the sugar can be easily regenerated by the action of carbon dioxide. Strontium monosulfide, SrS, is employed as a depilatory and as an ingredient in phosphors for electroluminescent devices and luminous paints.
Strontium ferrites comprise a family of compounds of general formula SrFexOy, formed from the high-temperature (1,000–1,300 °C, or 1,800–2,400 °F) reaction between SrCO3 and Fe2O3. Permanent ceramic magnets are made from strontium ferrites and find use in applications as diverse as loudspeakers, motors for automobile windshield wipers, and children’s toys.
Additional Information:
Appearance
A soft, silvery metal that burns in air and reacts with water.
Uses
Strontium is best known for the brilliant reds its salts give to fireworks and flares. It is also used in producing ferrite magnets and refining zinc.
Modern ‘glow-in-the-dark’ paints and plastics contain strontium aluminate. They absorb light during the day and release it slowly for hours afterwards.
Strontium-90, a radioactive isotope, is a by-product of nuclear reactors and present in nuclear fallout. It has a half-life of 28 years. It is absorbed by bone tissue instead of calcium and can destroy bone marrow and cause cancer. However, it is also useful as it is one of the best high-energy beta-emitters known. It can be used to generate electricity for space vehicles, remote weather stations and navigation buoys. It can also be used for thickness gauges and to remove static charges from machinery handling paper or plastic.
Strontium chloride hexahydrate is an ingredient in toothpaste for sensitive teeth.
Biological role
Strontium is incorporated into the shells of some deep-sea creatures and is essential to some stony corals. It has no biological role in humans and is non-toxic. Because it is similar to calcium, it can mimic its way into our bodies, ending up in our bones.
Radioactive strontium-90, which is produced in nuclear explosions and released during nuclear plant accidents, is particularly dangerous because it can be absorbed into the bones of young children.
Natural abundance
Strontium is found mainly in the minerals celestite and strontianite. China is now the leading producer of strontium. Strontium metal can be prepared by electrolysis of the molten strontium chloride and potassium chloride, or by reducing strontium oxide with aluminium in a vacuum.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1
- Index
- » Science HQ
- » Strontium